Kisspeptin is arguably the most important reproductive neuropeptide discovered in the 21st century. Since the landmark 2003 finding that mutations in its receptor cause complete reproductive failure, kisspeptin has reshaped our understanding of how the brain controls fertility — and opened doors to novel treatments for infertility, hormonal disorders, and potentially cancer.
This article covers the science: how kisspeptin works, what the clinical research shows, and where the field is heading. For practical dosing protocols and administration guidance, see our kisspeptin dosing guide.
The Discovery: From Cancer Lab to Reproductive Revolution
Kisspeptin has one of the most unusual origin stories in endocrinology.
In 1996, researchers at Penn State identified a gene that suppressed metastasis in melanoma cells. They named it KiSS-1 — a nod to Hershey's Kisses (the lab was in Hershey, Pennsylvania) and an acronym for "KiSS" (where SS stood for "suppressor sequence"). The protein product was dubbed "metastin" for its metastasis-suppressing properties (Lee et al., 1996).
For seven years, KISS1 was studied purely as a tumor suppressor. Then in 2003, two independent groups made a breakthrough: mutations in GPR54 — the receptor for kisspeptin — caused hypogonadotropic hypogonadism in both humans and mice. Patients with loss-of-function GPR54 mutations failed to enter puberty and were completely infertile (de Roux et al., 2003; Seminara et al., 2003).
This was the eureka moment: kisspeptin wasn't just a cancer gene — it was the master switch of reproduction.
Mechanism of Action: The GnRH-LH/FSH Cascade
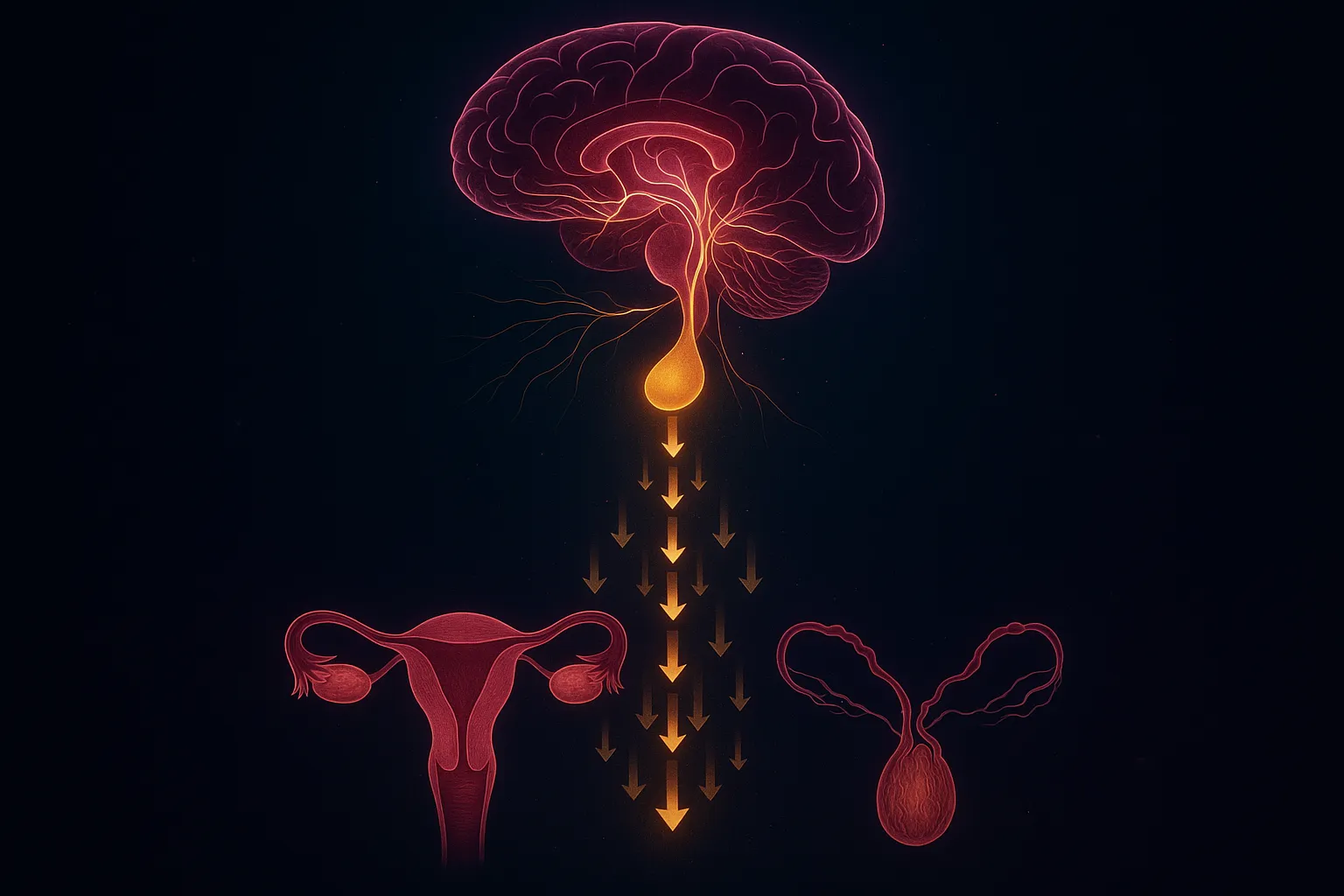
Kisspeptin sits at the very top of the hypothalamic-pituitary-gonadal (HPG) axis. Understanding this cascade explains virtually all of its clinical effects.
The Signaling Chain
- Kisspeptin neurons fire in the arcuate nucleus and anteroventral periventricular nucleus (AVPV) of the hypothalamus
- Kisspeptin binds KISS1R (GPR54) on GnRH neuron cell bodies and terminals
- GnRH is released in pulses into the hypophyseal portal system
- GnRH activates pituitary gonadotrophs → secretion of LH (luteinizing hormone) and FSH (follicle-stimulating hormone)
- LH and FSH act on the gonads → testosterone production in males, follicular development and ovulation in females
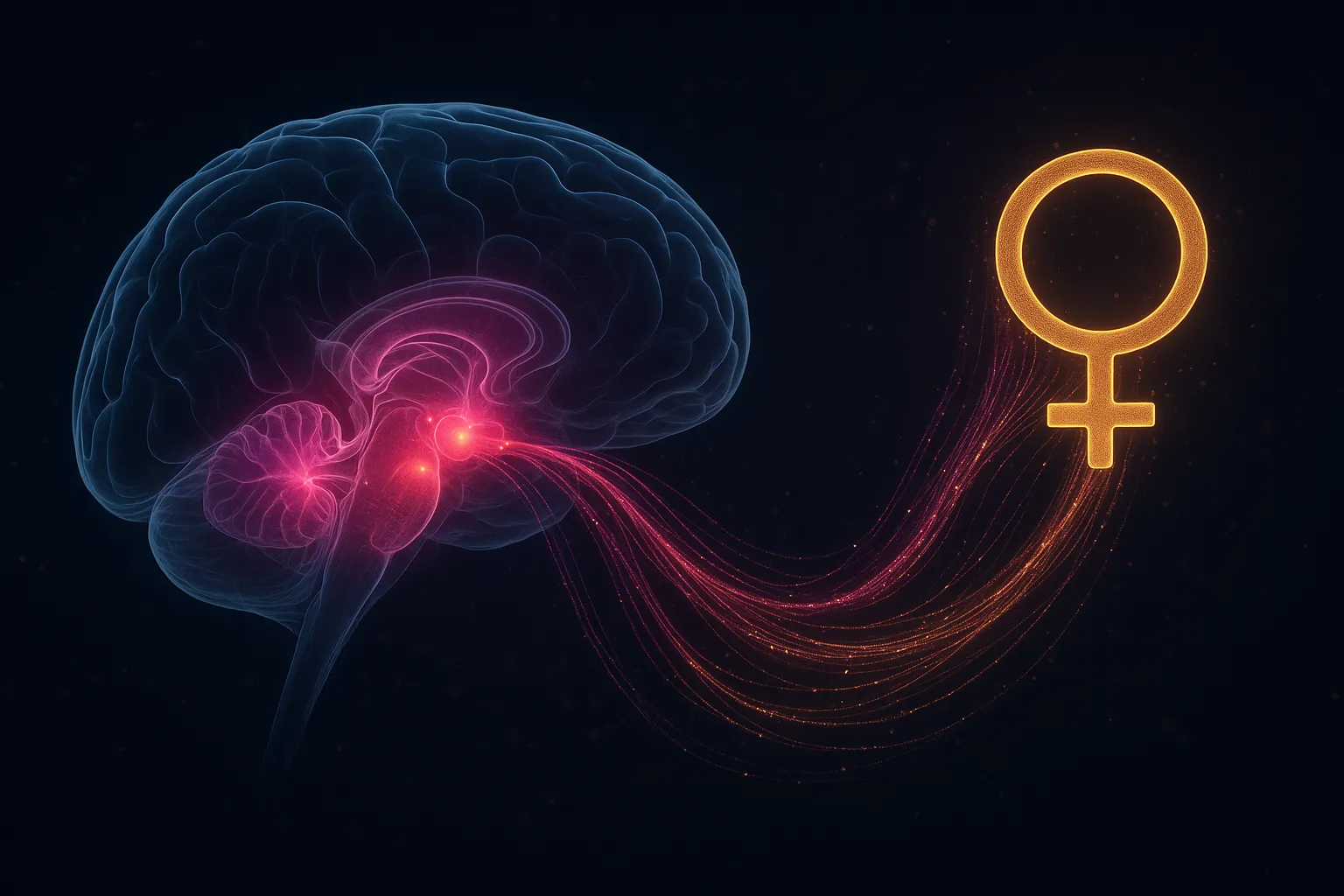
Why Kisspeptin Is Called the "Gatekeeper"
Kisspeptin neurons integrate signals from across the body — sex steroid feedback, metabolic status (leptin, insulin), circadian rhythms, stress hormones, and seasonal cues. They're the convergence point where the brain decides: is it safe and appropriate to reproduce right now?
This is why kisspeptin dysfunction doesn't just reduce fertility — it eliminates it entirely. Without kisspeptin signaling, the entire reproductive axis goes silent.
Pulsatile vs Continuous Signaling
A critical feature: kisspeptin neurons in the arcuate nucleus function as the GnRH pulse generator. They fire in coordinated bursts approximately every 60–90 minutes, with each pulse driving a corresponding GnRH pulse.
This pulsatility is not optional — it's required. Continuous GnRH exposure (as with GnRH agonist drugs like leuprolide) initially stimulates but then suppresses the pituitary through receptor desensitization. The same principle applies to kisspeptin: continuous exposure desensitizes KISS1R. This has direct implications for dosing — see our kisspeptin dosing guide for pulsatile protocols.
Fertility Applications
Kisspeptin's most advanced clinical applications are in reproductive medicine.
IVF: A Safer Oocyte Maturation Trigger
In standard IVF protocols, human chorionic gonadotropin (hCG) is used to trigger final oocyte maturation before egg retrieval. But hCG carries a significant risk of ovarian hyperstimulation syndrome (OHSS) — a potentially life-threatening complication in women with high ovarian response.
Kisspeptin-54 offers a fundamentally different approach. Because kisspeptin triggers an endogenous LH surge (rather than directly mimicking LH like hCG), the resulting hormonal response is more physiological and self-limiting.
Key clinical findings:
- Jayasena et al. (2014) demonstrated that a single subcutaneous injection of kisspeptin-54 successfully triggered oocyte maturation in IVF patients, with egg fertilization, embryo development, and pregnancies achieved (PMID: 25036713)
- Abbara et al. (2015) showed kisspeptin-54 effectively triggered oocyte maturation in women at high risk of OHSS, with zero cases of OHSS in the kisspeptin group (PMID: 26192876)
- The kisspeptin-triggered LH surge is shorter and more physiological than hCG-induced stimulation, which may explain the reduced OHSS risk
This represents a potential paradigm shift in IVF safety — particularly for women with polycystic ovary syndrome (PCOS) or high antral follicle counts who are most vulnerable to OHSS.
Hypothalamic Amenorrhea
Women with hypothalamic amenorrhea (absent periods due to stress, low body weight, or excessive exercise) have suppressed kisspeptin signaling. Exogenous kisspeptin can restart the HPG axis in these patients.
Clinical studies have shown that kisspeptin-54 infusion restores LH pulsatility in women with hypothalamic amenorrhea, providing proof of concept for kisspeptin-based treatments for this common condition.
Male Hypogonadotropic Hypogonadism
In men with central hypogonadism (low testosterone due to inadequate pituitary LH/FSH), kisspeptin represents a potential alternative to testosterone replacement or hCG therapy.
The first human kisspeptin study demonstrated that kisspeptin-54 infusion in healthy men produced robust, dose-dependent increases in LH, FSH, and testosterone (Dhillo et al., 2005). Subsequent studies confirmed these effects via subcutaneous administration.
The advantage over testosterone replacement: kisspeptin preserves the entire HPG axis, maintaining endogenous testosterone production, spermatogenesis, and testicular function — rather than shutting them down through negative feedback.
Sexual Function and Brain Effects
Beyond its hormonal effects, kisspeptin has direct actions on brain circuits involved in sexual arousal and emotional processing.
The fMRI Evidence
A landmark 2017 randomized controlled trial used functional MRI to study kisspeptin's brain effects in healthy men. Key findings:
- Kisspeptin-54 infusion enhanced limbic brain activity specifically in response to sexual stimuli — particularly in the amygdala, cingulate cortex, and globus pallidus
- This enhancement correlated with psychometric measures of sexual arousal, reward, and reduced sexual aversion
- Kisspeptin also modulated brain responses to couple-bonding stimuli (romantic, non-sexual images), suggesting effects on pair bonding beyond pure sexual arousal
- (Comninos et al., 2017)
HSDD Clinical Trial (2023)
A randomized clinical trial in men with hypoactive sexual desire disorder (HSDD) found that kisspeptin administration:
- Enhanced brain activity in sexual processing regions on fMRI
- Increased penile tumescence in response to sexual stimuli
- Improved behavioral measures of sexual desire
- Was well-tolerated with no serious adverse events
- (Ertl et al., 2023)
This is particularly significant because it demonstrates kisspeptin's effects in a clinical population with sexual dysfunction — not just healthy volunteers. It also distinguishes kisspeptin from PT-141, which acts through a completely different mechanism (melanocortin MC4R activation vs hormonal/limbic modulation). For a detailed comparison, see our kisspeptin vs PT-141 analysis.
Emerging research reveals that kisspeptin signaling extends beyond reproduction into metabolic regulation — which makes biological sense, since reproduction is energetically expensive and should be suppressed when metabolic resources are scarce.
Body Weight and Energy Expenditure
Tolson et al. (2014) demonstrated that Kiss1r knockout mice (lacking the kisspeptin receptor) developed:
- Significant obesity — particularly in females
- Decreased metabolic rate and energy expenditure
- Glucose intolerance and impaired insulin sensitivity
- These effects were partially independent of gonadal hormones, suggesting direct metabolic roles for kisspeptin signaling
- (PMID: 24937427)
Appetite and Feeding Regulation
Kisspeptin neurons interact with other hypothalamic systems involved in energy balance, including:
- Leptin signaling — leptin (the satiety hormone from fat tissue) acts partly through kisspeptin neurons to communicate metabolic status to the reproductive axis
- NPY/AgRP neurons — these hunger-promoting neurons interact with kisspeptin circuits in the arcuate nucleus
- Brown adipose tissue (BAT) — KISS1R is expressed in BAT, and conditional knockout of kisspeptin signaling in BAT altered metabolic rate in mice
This metabolic dimension is still in early research stages. No clinical metabolic applications exist for kisspeptin, but the connection between reproductive and metabolic signaling through kisspeptin neurons is an active research area.
Tumor Suppression: The KISS1 Legacy
Kisspeptin's original discovery was as a metastasis suppressor — and this cancer biology remains relevant.
The KISS1 Gene in Cancer
The KISS1 gene was identified because its expression was dramatically reduced in metastatic melanoma cells compared to non-metastatic cells. When KISS1 was re-introduced into metastatic cell lines, their ability to metastasize was suppressed by over 95% — without affecting primary tumor growth (Lee et al., 1996).
Key cancer-related findings:
- KISS1 suppresses metastasis, not primary tumor growth — it specifically inhibits the ability of cancer cells to establish secondary tumors
- Mechanism: kisspeptin/KISS1R signaling inhibits matrix metalloproteinase (MMP) activity and reduces cancer cell migration and invasion
- Multiple cancer types: reduced KISS1 expression has been associated with metastatic progression in melanoma, breast cancer, ovarian cancer, bladder cancer, gastric cancer, and pancreatic cancer
- Prognostic marker: low KISS1 expression in tumor biopsies often correlates with worse metastatic outcomes
Limitations
- KISS1's tumor-suppressive role is primarily demonstrated in cell lines and animal models
- No clinical trials have used kisspeptin as a cancer treatment
- The relationship between exogenous kisspeptin peptide administration and endogenous KISS1 gene expression in tumors is complex and not directly translatable
- Kisspeptin's stimulation of sex steroids could theoretically promote hormone-sensitive cancers — creating a paradox with its metastasis-suppressing properties
Who Is Kisspeptin Research Relevant For?
Based on the current evidence, kisspeptin research is most relevant for:
- Women undergoing IVF — particularly those at high risk of OHSS, where kisspeptin-54 as an oocyte maturation trigger has the strongest clinical evidence
- Men and women with hypogonadotropic hypogonadism — kisspeptin can restart the HPG axis at the highest level, preserving natural hormone production
- Women with hypothalamic amenorrhea — kisspeptin can restore LH pulsatility in this functional hypothalamic disorder
- Researchers studying sexual dysfunction — the fMRI and HSDD trial data suggest a novel approach to sexual desire disorders distinct from existing therapies
- Cancer biology researchers — KISS1/kisspeptin as a metastasis suppressor remains an active research field
Key Takeaways
- Kisspeptin is the master upstream regulator of the reproductive hormone axis — it controls GnRH, which controls everything downstream
- IVF applications are the most clinically advanced — kisspeptin-54 as an OHSS-free oocyte maturation trigger is a genuine breakthrough
- Brain effects extend beyond hormones — kisspeptin modulates sexual arousal and emotional processing via direct limbic actions
- Metabolic connections are real but early — kisspeptin links reproduction to energy balance, but clinical metabolic applications don't exist yet
- The cancer connection is foundational — KISS1 was discovered as a metastasis suppressor, and this biology remains relevant
- Pulsatile dosing matters — see our kisspeptin dosing guide for why continuous exposure backfires
For educational and research purposes only. This is not medical advice. Kisspeptin is not FDA-approved for any indication.


