The Next-Generation Weight Loss Battle
Retatrutide and Tirzepatide represent the cutting edge of metabolic medicine. Both surpass first-generation GLP-1 agonists, but they work through different mechanisms — dual vs triple receptor agonism.
This head-to-head comparison covers the real differences based on published clinical trial data.
Quick Verdict
For maximum weight loss: Retatrutide edges ahead with 24.2% weight loss vs tirzepatide's 22.5% at top doses.
For proven track record: Tirzepatide is FDA-approved and available now. Retatrutide is still in Phase 3 trials.
For mechanism: Retatrutide's triple agonism (GLP-1/GIP/Glucagon) adds fat-burning pathways that tirzepatide's dual agonism (GLP-1/GIP) lacks.
Mechanism Comparison
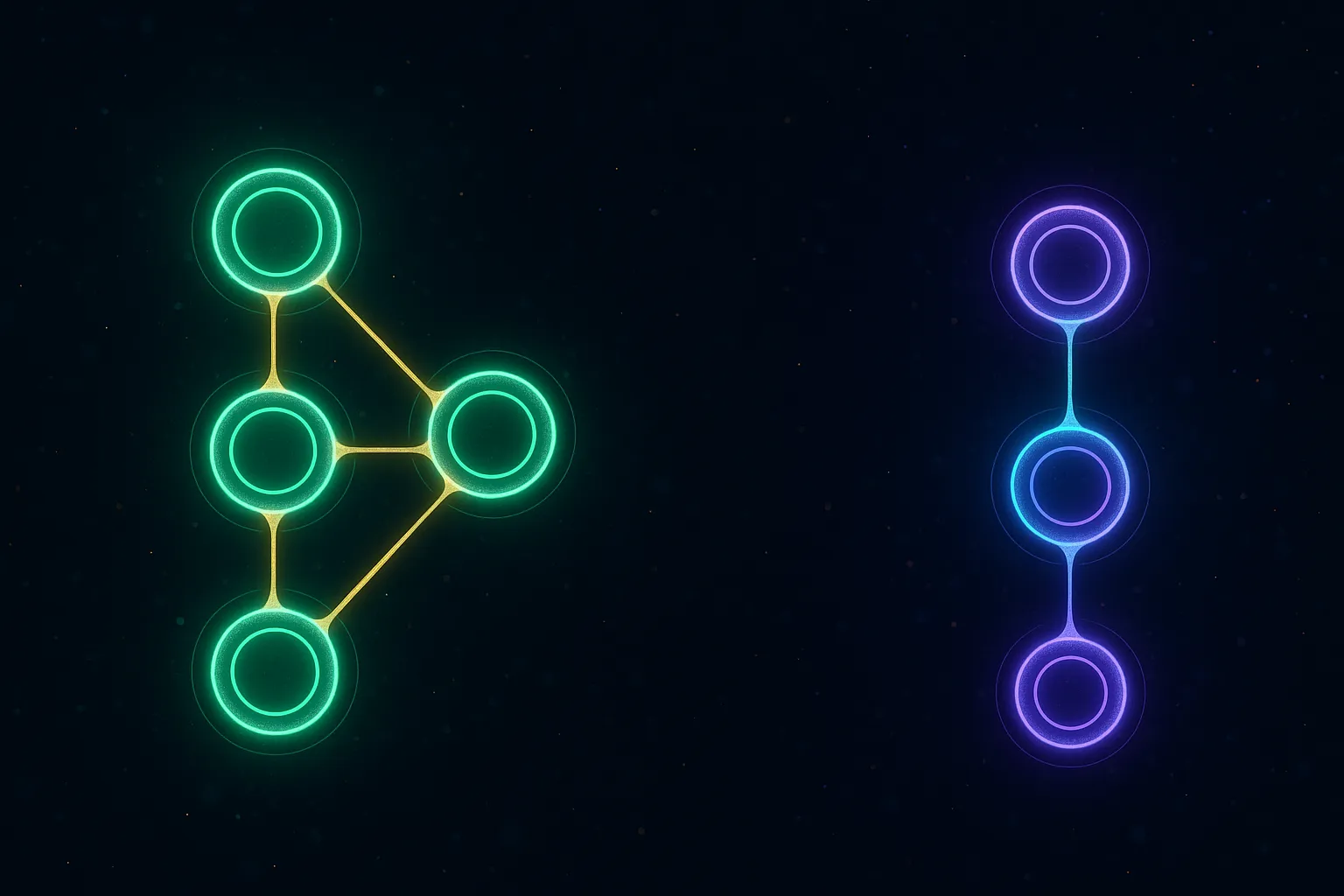
Tirzepatide (Dual GIP/GLP-1 Agonist)
Tirzepatide activates two complementary pathways:
- GLP-1 receptor — appetite suppression, delayed gastric emptying, improved insulin sensitivity
- GIP receptor — enhances insulin secretion and fat metabolism
- Synergistic effects — the dual mechanism produces greater metabolic benefits than single GLP-1 agonists
Retatrutide (Triple GLP-1/GIP/Glucagon Agonist)
Retatrutide adds a third target to tirzepatide's dual approach:
- GLP-1 + GIP effects — same appetite and metabolic benefits as tirzepatide
- Glucagon receptor activation — increases metabolic rate, enhances fat oxidation, promotes lipolysis
- Metabolic synergy — the triple mechanism creates the most comprehensive metabolic intervention available
The glucagon difference: Glucagon receptor agonism specifically targets fat burning and energy expenditure — pathways that GLP-1/GIP don't directly address.
Weight Loss Head-to-Head
Retatrutide Phase 2 Results (NEJM 2023)
The definitive Phase 2 trial studied retatrutide over 48 weeks in adults with obesity:
- Primary endpoint (24 weeks): Up to 17.5% weight loss (12 mg dose)
- Secondary endpoint (48 weeks): Up to 24.2% weight loss (12 mg dose)
- Dose-response: 1 mg (7.2%), 4 mg (12.9%), 8 mg (17.5%), 12 mg (24.2%)
- ≥15% responders: 83% achieved clinically significant weight loss at 12 mg
Tirzepatide SURMOUNT-1 Results (NEJM 2022)
The pivotal Phase 3 trial studied tirzepatide over 72 weeks in adults with obesity:
- Weight loss by dose: 5 mg (16.0%), 10 mg (21.4%), 15 mg (22.5%)
- ≥20% responders: 55% achieved substantial weight loss at 15 mg
- ≥25% responders: 36% achieved extreme weight loss at 15 mg
- Duration advantage: 72-week data vs retatrutide's 48-week data
Cross-Trial Comparison
Direct head-to-head trials haven't been conducted, but cross-trial analysis shows:
| Metric | Retatrutide (12 mg) | Tirzepatide (15 mg) |
|---|---|---|
| Peak weight loss | 24.2% at 48 weeks | 22.5% at 72 weeks |
| Trial duration | 48 weeks | 72 weeks |
| ≥20% responders | ~70% (estimated) | 55% |
| FDA status | Phase 3 trials | Approved |
Retatrutide advantage: Higher peak weight loss in shorter timeframe suggests superior efficacy.