
Tirzepatide is a dual GLP-1/GIP receptor agonist that provides superior weight loss and glycemic control compared to single-target GLP-1 agonists like semaglutide. FDA-approved for diabetes and weight management, with clinical data showing up to 22.5% weight loss.
FDA-approved for diabetes and weight management. Research peptide protocols use lower doses than pharmaceutical formulations. This is not medical advice.
Quick Reference: Standard Protocol
| Parameter |
Standard Protocol |
| Dose |
0.5 mg (10 units on insulin syringe) |
| Route |
Subcutaneous injection (abdomen or thigh) |
| Timing |
AM, empty stomach |
| Frequency |
3x per week (M/W/F or T/Th/S) |
| Cycle |
8 weeks on, 8 weeks off |
| Vial size |
10 mg |
| Reconstitution |
2 mL bacteriostatic water → 5,000 mcg/mL |
| Draw amount |
10 units on insulin syringe |
| Storage |
Refrigerate, use within 28 days |
Start at 0.25 mg 3x weekly for 2-4 weeks to assess tolerance, then increase to 0.5 mg 3x weekly. Split dosing provides more stable levels than once-weekly injection. For the full tirzepatide profile, vendor pricing, and stacking options, see our tirzepatide peptide page.
Cycling Details
The standard cycle is 8 weeks on, 8 weeks off. Start conservatively at 0.25 mg 3x weekly (0.75 mg total weekly) for the first 2-4 weeks — the dual GLP-1/GIP mechanism can cause more intense initial GI side effects than single-target agonists.
Increase to 0.5 mg 3x weekly (1.5 mg total weekly) if well-tolerated. The split dosing approach (3x weekly vs once weekly) maintains more consistent hormone levels and reduces peak-related side effects. Some users continue until goal weight, then cycle off.
Enhanced Protocol (FDA/Clinical)
Note: The standard protocol above uses research peptide community doses. The FDA-approved protocol below uses significantly higher doses.
| Parameter |
FDA Titration |
| Week 1-4 |
2.5 mg weekly |
| Week 5-8 |
5 mg weekly |
| Week 9-12 |
7.5 mg weekly |
| Week 13-16 |
10 mg weekly |
| Week 17-20 |
12.5 mg weekly |
| Maintenance |
Up to 15 mg weekly |
Community research peptide protocols use 1.5 mg/week total — roughly 60% of the FDA starting dose and 10% of the maximum. Lower doses still provide meaningful effects with substantially fewer side effects.
Routes of Administration
Subcutaneous (standard and only route): Rotate between abdomen, thigh, and upper arm. Abdomen typically provides the most consistent absorption. Use 29-31 gauge insulin syringe. Inject same time each dosing day, morning on empty stomach preferred.
Reconstitution Quick Reference
| Vial Size |
BAC Water |
Concentration |
0.25 mg |
0.5 mg |
| 10 mg |
2 mL |
5,000 mcg/mL |
5 units |
10 units |
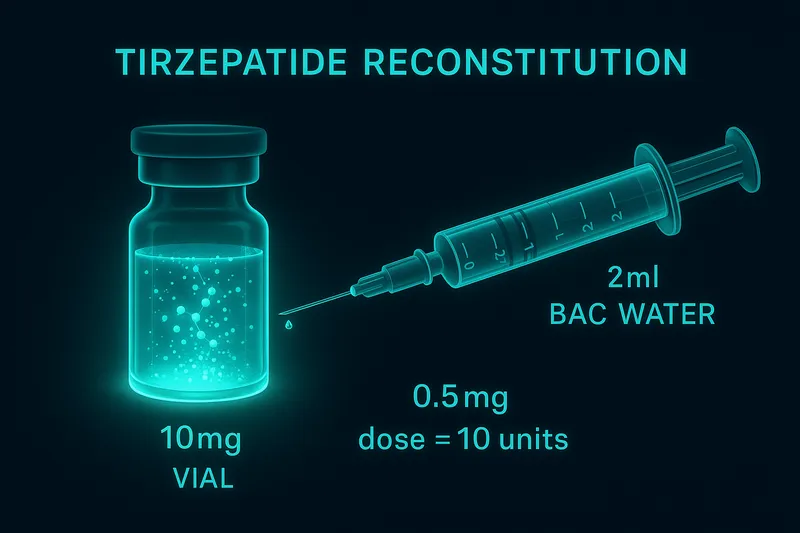
10 mg vial + 2 mL BAC water = 5,000 mcg/mL. Your 0.5 mg dose is 10 units on an insulin syringe.
Swirl gently — do not shake. Refrigerate at 2-8°C and use within 28 days.
Where These Numbers Come From
Community protocols use significantly lower doses than FDA-approved protocols, with good rationale.
SURMOUNT-1 Trial (Jastreboff et al., 2022): Even at the 2.5 mg starting dose, participants achieved 16% weight loss. The dual GLP-1/GIP mechanism creates potent metabolic effects at lower doses than single-target agonists.
Head-to-Head vs Semaglutide (Frias et al., 2021): Tirzepatide demonstrated superior glycemic control compared to semaglutide, supporting the dual-agonist advantage.
Research Peptide Variability: Purity and potency vary between suppliers. Starting conservatively accounts for this uncertainty. Individual sensitivity varies — some achieve excellent results well below maximum titration.
Stacking Protocols
Tirzepatide + AOD-9604
| Peptide |
Dose |
Route |
Timing |
Purpose |
| Tirzepatide |
0.5 mg 3x/week |
SC |
AM, empty stomach |
Appetite suppression, metabolic control |
| AOD-9604 |
300 mcg daily |
SC |
AM or pre-workout |
Direct lipolytic effects |
Tirzepatide + BPC-157
| Peptide |
Dose |
Route |
Timing |
Purpose |
| Tirzepatide |
0.5 mg 3x/week |
SC |
AM |
Weight loss, metabolic control |
| BPC-157 |
250-500 mcg daily |
SC |
AM or PM |
Tissue recovery, gut health |
Do NOT stack with: Other GLP-1 agonists (semaglutide, liraglutide) — overlapping mechanisms create unnecessary side effect risk.
Side Effects & Safety
- Nausea — up to 56% at higher FDA doses; much lower at community doses
- Vomiting — up to 36% at high doses
- Diarrhea — 23-26%
- Constipation — 17-19%
- Fatigue — 11-13%
- Pancreatitis risk — stop immediately if severe abdominal pain develops
- Gallbladder issues — rapid weight loss increases gallstone risk
- Thyroid C-cell tumors — same black box warning as semaglutide (animal studies)
GI side effects are more intense than semaglutide due to dual mechanism. Slow titration significantly reduces severity.
Frequently Asked Questions
What is the standard tirzepatide dose?
0.5 mg subcutaneous 3 times per week (1.5 mg total weekly) in the morning on an empty stomach, cycled 8 weeks on / 8 weeks off. Draw 10 units from a 10 mg vial reconstituted with 2 mL BAC water.
How do I dose tirzepatide 3x per week?
Inject 0.5 mg every other day — Monday/Wednesday/Friday or Tuesday/Thursday/Saturday. Split dosing provides more stable levels and fewer GI side effects than once-weekly.
How long should a tirzepatide cycle last?
8 weeks on, 8 weeks off, or continue until goal weight. Cycling helps maintain receptor sensitivity.
Should I take tirzepatide on an empty stomach?
Yes — morning, empty stomach. Wait 30-60 minutes before eating. The dual mechanism creates stronger gastric emptying delays.
Is tirzepatide FDA-approved?
Yes — for type 2 diabetes and chronic weight management at 2.5-15 mg weekly. Research peptide protocols use different (lower) doses.
How do I reconstitute tirzepatide?
Add 2 mL BAC water to a 10 mg vial (5,000 mcg/mL). 0.5 mg = 10 units on an insulin syringe. Swirl gently, refrigerate, use within 28 days.
References
| Citation |
Topic |
PMID |
| Jastreboff et al., N Engl J Med (2022) |
SURMOUNT-1: tirzepatide for weight management |
35658024 |
| Frias et al., N Engl J Med (2021) |
Tirzepatide vs semaglutide head-to-head |
34378548 |
| Samms et al., Nat Rev Drug Discov (2020) |
GIP receptor biology and therapeutic potential |
32004751 |
| Dahl et al., Diabetes Obes Metab (2022) |
SURMOUNT-2: tirzepatide in diabetes with obesity |
35441470 |
| Rosenstock et al., Lancet (2021) |
SURPASS-1: tirzepatide monotherapy in T2D |
34186022 |
For educational and research purposes only. This is not medical advice. Research peptides are not FDA-approved for human use.

