
Retatrutide is a triple-agonist peptide targeting GLP-1 + GIP + glucagon receptors simultaneously. Phase 2 trials showed up to 24.2% body weight loss in 48 weeks at higher doses — the most potent weight-loss data from any metabolic peptide studied to date.
Retatrutide is not FDA-approved. Everything below reflects published clinical trials and community protocols. This is not medical advice.
Quick Reference: Standard Protocol
| Parameter |
Detail |
| Vial |
10 mg |
| BAC Water |
2 mL |
| Concentration |
5 mg/mL |
| Dose |
0.5 mg (10 units on insulin syringe) |
| Route |
Subcutaneous |
| Timing |
AM, empty stomach |
| Frequency |
3x/week (Mon/Wed/Fri) |
| Cycle |
8 weeks on, 8 weeks off |
| Storage |
Refrigerate, use within 28 days |
Most people start at 0.25 mg for the first week to assess GI tolerance, then increase to 0.5 mg.
For the full retatrutide profile and vendor pricing, see our Retatrutide peptide page.
Cycling Details
The standard cycle is 0.5 mg 3x/week (1.5 mg/week total) for 8 weeks, then 8 weeks off. Some users run goal-oriented cycles — continuing until target weight loss, then cycling off. Maintenance phases may drop to 2x/week (1 mg/week total).
The 3x/week split (vs. single weekly injection used in clinical trials) may provide more stable receptor activation throughout the week.
Routes of Administration
Subcutaneous injection is the only route. Retatrutide cannot be taken orally.
- Sites: Abdomen, thigh, or anywhere with subcutaneous fat
- Volume: 0.1 mL with insulin syringe (29-31 gauge)
- Rotation: Rotate injection sites to prevent lipodystrophy
Reconstitution Quick Reference
| Vial Size |
BAC Water |
Concentration |
0.25 mg |
0.5 mg |
1 mg |
| 10 mg |
2 mL |
5 mg/mL |
5 units |
10 units |
20 units |
Math: 10 mg / 2 mL = 5 mg/mL. For 0.5 mg: 0.5 / 5 = 0.1 mL = 10 units.
Swirl gently — do not shake. Refrigerate at 2-8 C, use within 28 days. For step-by-step instructions and vial size comparisons, see the full Retatrutide Reconstitution Guide.
Where These Numbers Come From
Community doses represent a conservative approach compared to clinical trials.
Phase 2 obesity trial: Escalating weekly doses from 1 mg to 12 mg over 48 weeks. Maximum weight loss: 24.2% at 12 mg/week (Jastreboff et al., 2023).
| Timeframe |
Clinical Dose |
Weekly Total |
| Weeks 0-4 |
1 mg weekly |
1 mg/week |
| Weeks 4-8 |
4 mg weekly |
4 mg/week |
| Weeks 8-12 |
8 mg weekly |
8 mg/week |
| Weeks 12-48 |
12 mg weekly |
12 mg/week |
Phase 2 diabetes trial: Dose-dependent HbA1c reductions — minimum effective dose for meaningful glucose control was 4 mg/week (Rosenstock et al., 2023). Community protocols at 1.5 mg/week fall below this threshold.
Why community doses are lower: No FDA approval means conservative self-titration. Lower doses reduce GI side effects. The clinical trial starting dose (1 mg/week) is already close to the community protocol (1.5 mg/week split 3x). Many see significant effects at these lower levels.
Research Vial Sizing
| Vial Size |
Not a Single Dose |
Provides |
| 12 mg |
12 mg is the vial total |
24 doses of 0.5 mg |
| 24 mg |
24 mg is the vial total |
48 doses of 0.5 mg |
| 60 mg |
60 mg is the vial total |
120 doses of 0.5 mg |
Larger vials offer better per-mg pricing. Compare current vial options and verified vendors at Best Retatrutide Vendors.
Stacking Protocols
| Stack |
Purpose |
Protocol |
| Retatrutide + BPC-157 |
GI protection during GLP-1 use |
BPC-157 250-500 mcg/day alongside retatrutide |
Retatrutide already hits three major metabolic pathways, leaving few complementary targets. Adding other GLP-1 agonists (semaglutide, tirzepatide) is redundant and increases side effects. Stacking with other weight loss peptides is generally unnecessary.
Side Effects & Safety
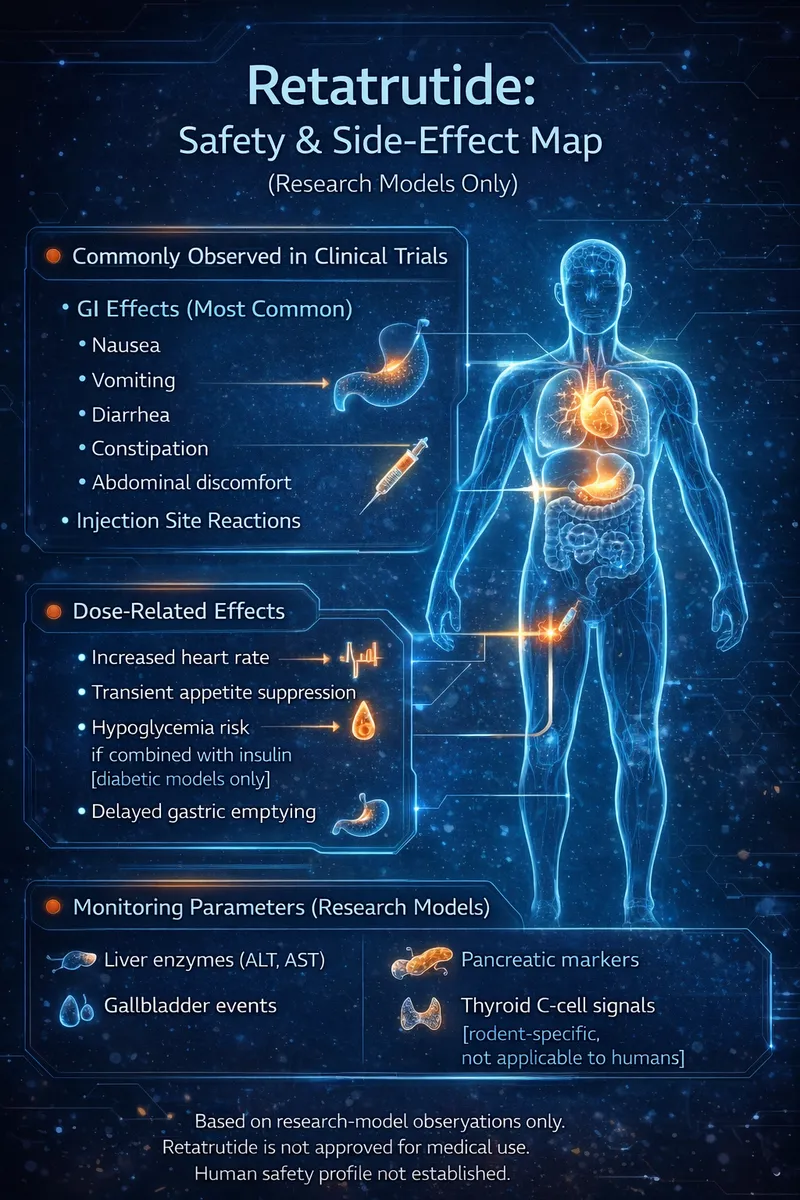
- Nausea — most common (~33% at high doses), dose-dependent, typically improves over time
- GI effects — vomiting, diarrhea, constipation, slowed gastric emptying (GLP-1 class effects)
- Heart rate changes — transient increases observed in trials, peaked at 24 weeks then declined
- Injection site reactions — mild redness or irritation
- Gallbladder risk — rapid weight loss increases gallstone risk (common with any rapid weight loss method)
- No major safety signals in 48-week trial up to 12 mg weekly
- At community doses (1.5 mg/week), side effects are notably milder than high-dose trial groups
Frequently Asked Questions
What is the standard retatrutide dose for weight loss?
0.5 mg subcutaneously 3 times per week (1.5 mg/week total), in the morning, cycled 8 weeks on / 8 weeks off. This is much lower than the clinical trial max of 12 mg/week.
Why are research vials sold as 12mg, 24mg, or 60mg?
These are total vial contents, not single doses. A 24 mg vial provides 48 doses at 0.5 mg each.
How does retatrutide compare to tirzepatide?
Based on Phase 2 data, retatrutide showed greater weight loss (24.2% max vs ~22% for tirzepatide). Retatrutide is a triple agonist (GLP-1 + GIP + glucagon) while tirzepatide is dual (GLP-1 + GIP).
Is retatrutide FDA-approved?
No — Phase 3 trials are ongoing. All dosing reflects research protocols and community use.
Should I start at 0.5mg or lower?
Many start at 0.25 mg for the first week to assess GI tolerance, then increase to 0.5 mg.
Can retatrutide be taken orally?
No — it's a peptide that would be destroyed by digestive enzymes. Subcutaneous injection only.
References
| Citation |
Topic |
PMID |
| Jastreboff AM, et al., N Engl J Med (2023) |
Phase 2 trial: Triple-hormone agonist for obesity |
37366315 |
| Rosenstock J, et al., Lancet (2023) |
Phase 2 trial: Retatrutide for type 2 diabetes |
37385280 |
This article is for educational and informational purposes only. It is not medical advice. Retatrutide is not FDA-approved for any indication. Consult a licensed healthcare provider before using any peptide.

