
Table of Contents
- What Is SS-31?
- How SS-31 Works (Mechanism of Action)
- Research Benefits at a Glance
- SS-31 vs Other Mitochondrial Peptides
- Dosing Overview
- Risks & Safety Considerations
- FAQ
- References
1. What Is SS-31?
SS-31 (also known as Elamipretide, Bendavia, and MTP-131) is a synthetic mitochondria-targeted tetrapeptide with the sequence D-Arg-Dmt-Lys-Phe-NH₂. It was developed by Hazel Szeto and Peter Bhatt at Weill Cornell Medical College and is named after the Szeto-Schiller (SS) peptide series.
Unlike most peptides in research, SS-31 doesn't bind a cell-surface receptor. Instead, it selectively concentrates in mitochondria — specifically at the inner mitochondrial membrane — where it interacts with cardiolipin, a phospholipid essential for electron transport chain (ETC) function.
SS-31 is found at especially high concentrations in:
- Inner mitochondrial membrane — where it binds cardiolipin at ETC complex interfaces
- Cardiac mitochondria — the tissue with the highest mitochondrial density
- Skeletal muscle mitochondria — critical for exercise capacity and recovery
- Renal mitochondria — relevant to ischemia-reperfusion injury models
- Retinal mitochondria — studied in age-related macular degeneration
What makes SS-31 unique among peptides is its mitochondrial targeting: the alternating aromatic-cationic motif (aromatic → cationic → aromatic → cationic) allows it to cross membranes and accumulate >1000-fold in mitochondria within minutes, driven by the mitochondrial membrane potential (Zhao et al., 2004).
Not to be confused with MOTS-c, which is an endogenous mitochondrial-derived peptide that acts primarily through AMPK activation and metabolic signaling rather than direct cardiolipin stabilization.
Why Is SS-31 Trending?
Search volume for SS-31 and Elamipretide has increased due to:
- Active Phase II/III clinical trials for heart failure (Barth syndrome, primary mitochondrial myopathy)
- Growing recognition that mitochondrial dysfunction underlies aging, heart disease, and neurodegeneration
- The unique mechanism — direct cardiolipin stabilization — which no other compound targets
- Published data showing reversal of age-related mitochondrial decline in animal models
- Interest in longevity peptides alongside Epitalon, MOTS-c, and Humanin
2. How SS-31 Works (Mechanism of Action)
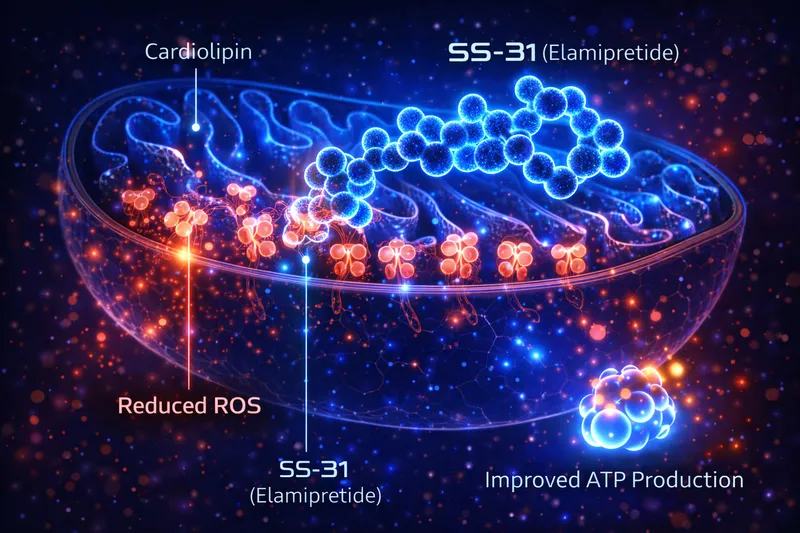
SS-31's biological effects are driven by a single, well-defined primary interaction — cardiolipin binding — that produces multiple downstream effects. This makes its mechanism unusually clean and well-characterized compared to multi-target peptides.
2.1 Cardiolipin Stabilization (Core Mechanism)
This is SS-31's defining mechanism and the reason it was developed.
Cardiolipin is a unique phospholipid found almost exclusively in the inner mitochondrial membrane. It is essential for:
- Anchoring electron transport chain (ETC) complexes I, III, IV, and V
- Maintaining cristae structure (the folds of the inner membrane)
- Enabling supercomplex formation (respiratory chain supercomplexes)
- Facilitating cytochrome c binding and electron transfer
SS-31 binds cardiolipin through electrostatic and hydrophobic interactions, stabilizing its structure and preventing peroxidation:
SS-31 → Binds cardiolipin → Stabilizes ETC complex interactions → ↑ Electron transfer efficiency → ↑ ATP production → ↓ Electron leak → ↓ ROS
Birk et al. demonstrated that SS-31 interacts selectively with cardiolipin, improving mitochondrial coupling and ATP synthesis while reducing reactive oxygen species (ROS) generation — the first direct evidence of cardiolipin-targeted therapy (Birk et al., 2013).
When cardiolipin is damaged (by oxidation, aging, or ischemia), ETC complexes destabilize, electron leak increases, ROS production rises, and ATP output falls. SS-31 directly reverses this cascade by restoring cardiolipin's structural role.
2.2 Electron Transport Chain Optimization
By stabilizing cardiolipin, SS-31 improves the efficiency of oxidative phosphorylation — the primary pathway cells use to produce ATP.
Key effects on ETC function:
- Improved Complex I–III–IV supercomplex stability — prevents disassembly that occurs with cardiolipin peroxidation
- Enhanced cytochrome c interaction — optimizes electron transfer between Complex III and IV
- Increased ATP/O₂ ratio — more ATP produced per oxygen consumed (improved coupling)
- Reduced proton leak — less energy wasted as heat
Siegel et al. showed that SS-31 restores mitochondrial ADP sensitivity in aged muscle — meaning aged mitochondria treated with SS-31 responded to energy demand like young mitochondria (Siegel et al., 2013). A 2023 follow-up confirmed SS-31 improves ADP sensitivity in aging human skeletal muscle mitochondria (Siegel et al., 2023).
2.3 ROS Reduction (Antioxidant Effect)
SS-31 is often described as a "mitochondrial antioxidant," but this is somewhat misleading. SS-31 is not a free radical scavenger in the traditional sense (like vitamin C or glutathione). Instead, it reduces ROS production at the source by preventing electron leak from the ETC.
This distinction is critical:
- Traditional antioxidants → mop up ROS after they're produced
- SS-31 → prevents excess ROS from being generated in the first place
By stabilizing cardiolipin and maintaining ETC efficiency, SS-31 reduces the electron leak that generates superoxide radicals at Complexes I and III. Szeto demonstrated this "upstream" antioxidant mechanism in a comprehensive review of the SS peptide series (Szeto, 2014).
Effects:
- Reduced mitochondrial superoxide — less oxidative damage to mitochondrial DNA and proteins
- Preserved mitochondrial membrane potential — maintains the driving force for ATP synthesis
- Reduced lipid peroxidation — protects cardiolipin and other membrane lipids
- Preserved NAD+/NADH ratio — maintains cellular redox balance
2.4 Anti-Apoptotic & Cytoprotective Signaling
Damaged mitochondria trigger apoptosis (programmed cell death) through cytochrome c release. SS-31 prevents this cascade by maintaining mitochondrial integrity:
- Prevents cytochrome c release from the intermembrane space
- Stabilizes mitochondrial membrane potential (ΔΨm)
- Reduces mitochondrial permeability transition pore (mPTP) opening — the "point of no return" for mitochondrial-driven cell death
Zhao et al. demonstrated SS-31's cytoprotective effects in ischemia-reperfusion models, showing reduced infarct size and improved cell survival in cardiac tissue (Zhao et al., 2004).
Mechanism Summary
| Pathway |
Primary Action |
Key Downstream Effect |
| Cardiolipin binding |
Stabilizes inner membrane phospholipid |
ETC complex stability |
| ETC optimization |
Improved supercomplex assembly |
↑ ATP production, ↑ coupling efficiency |
| ROS reduction |
↓ Electron leak at source |
↓ Oxidative damage to mitochondrial components |
| Cytoprotection |
Prevents cytochrome c release |
↓ Apoptosis, ↑ cell survival |
3. Research Benefits at a Glance
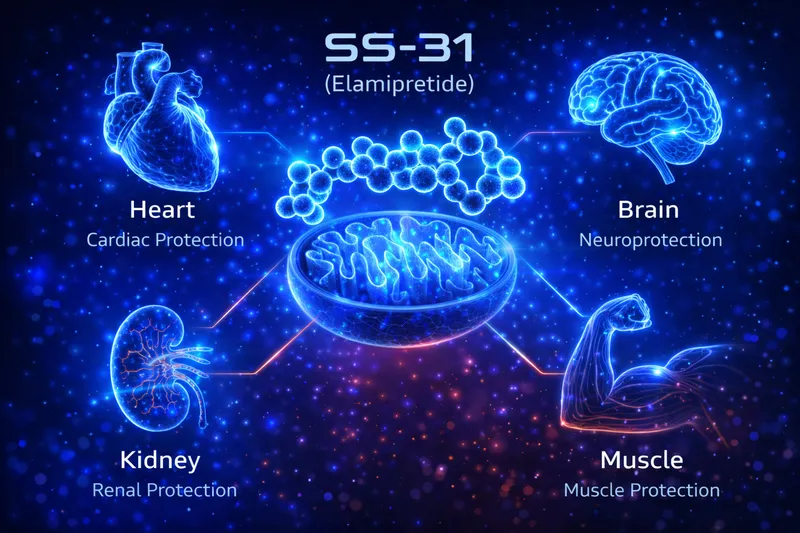
SS-31 research spans tissues with high mitochondrial demand — heart, skeletal muscle, kidney, brain, and retina. Below is a brief overview of documented effects. For the full dosing protocols used in these studies, see our SS-31 Dosing Guide.
-
Cardiac Protection & Heart Failure — The most clinically advanced application. SS-31 improved left ventricular function, reduced infarct size, and prevented cardiac remodeling in heart failure models. Phase II trials in Barth syndrome (a genetic cardiolipin deficiency) showed improved 6-minute walk test distance (Thompson et al., 2021). The TAZPOWER and PROGRESS-HF trials evaluated SS-31 in heart failure with reduced ejection fraction.
-
Skeletal Muscle & Exercise Capacity — SS-31 reversed age-related decline in mitochondrial ADP sensitivity, restored ATP production, and improved exercise tolerance in aged mice. Campbell et al. showed that even short-term SS-31 treatment restored skeletal muscle mitochondrial function to near-youthful levels (Campbell et al., 2019).
-
Aging & Longevity — SS-31 reversed age-related mitochondrial dysfunction across multiple tissues. Siegel et al. demonstrated that SS-31 rapidly reverses age-related redox changes and restores ADP-stimulated respiration in skeletal muscle mitochondria (Siegel et al., 2013). This positions SS-31 as one of the most studied anti-aging peptides alongside Epitalon.
-
Kidney Ischemia-Reperfusion Injury — SS-31 reduced oxidative damage and preserved renal function in ischemia-reperfusion models, protecting mitochondria during the critical reperfusion window when ROS generation peaks (Szeto et al., 2011).
-
Neurodegenerative Disease Models — Emerging research shows SS-31 protects neuronal mitochondria in Alzheimer's, Parkinson's, and ALS models. Manczak et al. demonstrated SS-31 reduced mitochondrial dysfunction, oxidative stress, and synaptic deterioration in an Alzheimer's disease mouse model (Manczak et al., 2010).
-
Retinal & Optic Nerve Protection — SS-31 protected retinal ganglion cells and preserved visual function in glaucoma and optic neuropathy models by maintaining mitochondrial integrity in the metabolically demanding retina.
→ Read the complete SS-31 dosing guide with protocols & study citations →
4. SS-31 vs Other Mitochondrial Peptides
SS-31 belongs to a growing class of mitochondrial peptides, but each works through fundamentally different mechanisms.
| Feature |
SS-31 (Elamipretide) |
MOTS-c |
Humanin |
| Origin |
Synthetic (Szeto-Schiller series) |
Endogenous (mtDNA-encoded) |
Endogenous (mtDNA-encoded) |
| Size |
4 amino acids |
16 amino acids |
24 amino acids |
| Target |
Inner mitochondrial membrane (cardiolipin) |
AMPK pathway (cytoplasmic/nuclear) |
IGFBP-3 / BAX (extracellular/cytoplasmic) |
| Primary mechanism |
Cardiolipin stabilization → ETC optimization |
AMPK activation → metabolic regulation |
Anti-apoptotic signaling → cytoprotection |
| Best studied for |
Heart failure, aging, ischemia-reperfusion |
Metabolic syndrome, exercise, insulin sensitivity |
Neurodegeneration, insulin resistance, aging |
| Clinical trials |
Phase II/III (heart failure, Barth syndrome) |
Preclinical |
Preclinical |
Key Distinction
SS-31 works inside mitochondria at the biophysical level (membrane lipid stabilization). MOTS-c and Humanin are signaling peptides that work through receptor-mediated and kinase pathways outside mitochondria, despite being encoded by mitochondrial DNA.
This means SS-31 and MOTS-c address mitochondrial dysfunction from completely different angles — making them mechanistically non-overlapping. For a detailed head-to-head comparison, see our SS-31 vs MOTS-c comparison.
5. Dosing Overview
For educational and research discussion only. This is not medical advice.
SS-31 has been evaluated in human clinical trials, providing more dosing data than most research peptides. Published protocols use subcutaneous and intravenous routes, with doses ranging from 0.01 to 0.25 mg/kg in clinical settings. The Phase II TAZPOWER trial used 40 mg SC once daily as the primary dose in Barth syndrome patients.
In preclinical models, doses of 0.1–3 mg/kg SC or IP are common, with effects observed as early as 1 hour post-injection due to rapid mitochondrial accumulation.
→ Read the complete SS-31 dosing guide with protocols, injection routes & study citations →
6. Risks & Safety Considerations
SS-31 has a favorable safety profile across multiple clinical trials — a significant advantage over peptides with only preclinical data.
6.1 Clinical Trial Safety Data
- Phase I: Single and multiple ascending IV doses (0.01–0.25 mg/kg) in healthy volunteers — well tolerated, no serious adverse events, no dose-limiting toxicity (Szeto, 2014)
- Phase II (TAZPOWER): 40 mg SC daily for 12 weeks in Barth syndrome — generally well tolerated (Thompson et al., 2021)
- Phase II (PROGRESS-HF): Evaluated in heart failure with reduced ejection fraction — safety endpoints met
6.2 Common Adverse Events
Across clinical trials, the most frequently reported adverse events are:
- Injection site reactions — mild pain, redness, or induration at SC injection sites
- Headache — mild, transient
- Nausea — typically mild and self-resolving
6.3 Limitations
- Long-term safety data (>12 months) is limited
- Efficacy has not been definitively established — Phase III trials for some indications have had mixed results
- The Barth syndrome trial (TAZPOWER) did not meet its primary endpoint (6-minute walk test), though secondary endpoints showed trends toward improvement
- Effects on healthy mitochondria at supraphysiological doses are not fully characterized
6.4 Theoretical Considerations
SS-31 concentrates rapidly in mitochondria, driven by membrane potential. Tissues with damaged mitochondria (reduced ΔΨm) may accumulate less SS-31 — potentially limiting efficacy in the most severely affected cells. However, this has not been a clinically significant issue in published trials.
FAQ
What is SS-31?
SS-31 (Elamipretide) is a synthetic mitochondria-targeted tetrapeptide that binds cardiolipin in the inner mitochondrial membrane, stabilizing electron transport chain function, increasing ATP production, and reducing oxidative stress.
Is SS-31 the same as Elamipretide?
Yes. SS-31, Elamipretide, Bendavia, and MTP-131 are all names for the same peptide: D-Arg-Dmt-Lys-Phe-NH₂. SS-31 is the research designation, Elamipretide is the clinical name.
How is SS-31 different from MOTS-c?
SS-31 works inside mitochondria by stabilizing cardiolipin at the inner membrane. MOTS-c is an endogenous mitochondrial-derived peptide that works through AMPK activation in the cytoplasm. They are mechanistically non-overlapping. See our SS-31 vs MOTS-c comparison for the full breakdown.
Has SS-31 been tested in humans?
Yes. SS-31 is one of the few mitochondrial peptides with Phase II/III clinical trial data. It has been evaluated in healthy volunteers, Barth syndrome, heart failure, and primary mitochondrial myopathy.
Where can I read about SS-31 dosing?
See our dedicated SS-31 Dosing Guide for clinical and preclinical dosing protocols, injection routes, and study citations.
- SS-31 Dosing Guide — Clinical and preclinical dosing protocols, injection routes, and study durations
- SS-31 vs MOTS-c — Two mitochondrial peptides, completely different mechanisms
- MOTS-c — Endogenous mitochondrial peptide for metabolic health
- Epitalon — Telomerase activator for cellular longevity
- Humanin — Cytoprotective peptide for neuroprotection
- Peptide Stacking Guide — How to combine peptides for complementary coverage
References
-
Zhao K, Zhao GM, Wu D, et al. "Cell-permeable peptide antioxidants targeted to inner mitochondrial membrane inhibit mitochondrial swelling, oxidative cell death, and reperfusion injury." J Biol Chem. 2004;279(33):34682-90. PubMed
-
Birk AV, Liu S, Soong Y, et al. "The mitochondrial-targeted compound SS-31 re-energizes ischemic mitochondria by interacting with cardiolipin." J Am Soc Nephrol. 2013;24(8):1250-61. PubMed
-
Siegel MP, Kruse SE, Percival JM, et al. "Mitochondrial-targeted peptide rapidly improves mitochondrial energetics and skeletal muscle performance in aged mice." Aging Cell. 2013;12(5):763-71. PubMed
-
Siegel MP, Wilber EL, Goh J, et al. "SS-31 improves ADP sensitivity in aged skeletal muscle mitochondria." GeroScience. 2023;45(4):2735-2745. PubMed
-
Szeto HH. "First-in-class cardiolipin-protective compound as a therapeutic agent to restore mitochondrial bioenergetics." Br J Pharmacol. 2014;171(8):2029-50. PubMed
This article is for educational and research purposes only. It is not medical advice.


