
Two Mitochondrial Peptides, Two Completely Different Approaches
SS-31 and MOTS-c are both classified as mitochondrial peptides — but that's where the similarity ends. They work through fundamentally different mechanisms, target different biological pathways, and have very different research profiles.
This comparison breaks down exactly how they differ and where each one has the strongest research support.
Quick Comparison
| Feature |
SS-31 (Elamipretide) |
MOTS-c |
| Type |
Synthetic tetrapeptide |
Endogenous mitochondrial-derived peptide |
| Size |
4 amino acids (D-Arg-Dmt-Lys-Phe-NH₂) |
16 amino acids |
| Origin |
Lab-designed (Szeto-Schiller series) |
Encoded by mitochondrial DNA (12S rRNA) |
| Primary target |
Cardiolipin (inner mitochondrial membrane) |
AMPK pathway (cytoplasmic/nuclear) |
| Where it works |
Inside mitochondria |
Outside mitochondria (cytoplasm → nucleus) |
| Primary effect |
↑ ATP production, ↓ ROS |
↑ Metabolic regulation, ↑ insulin sensitivity |
| Best studied for |
Heart failure, aging, ischemia |
Metabolic syndrome, obesity, exercise |
| Clinical trials |
Phase II/III (Barth syndrome, HF) |
None (preclinical only) |
| Onset |
Minutes (rapid mitochondrial uptake) |
Hours to days (gene expression changes) |
Mechanism of Action
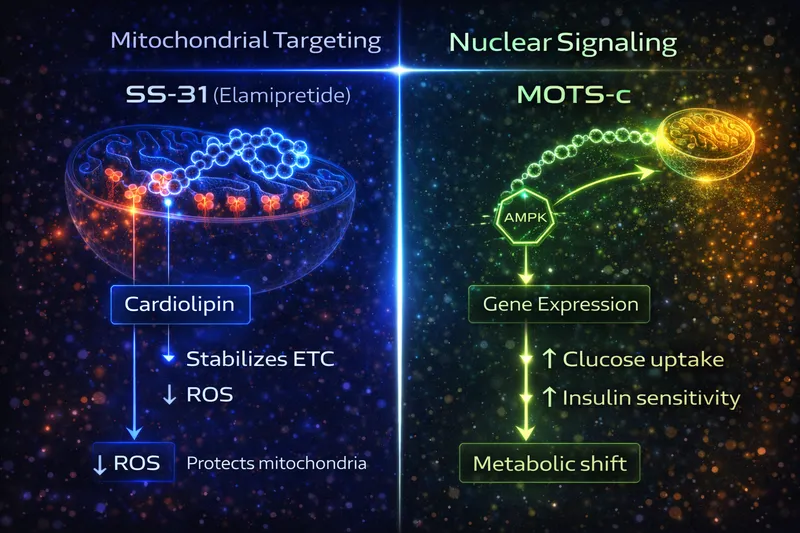
SS-31: Cardiolipin Stabilization (Biophysical)
SS-31 works at the biophysical level — it doesn't activate a receptor or signaling cascade. Instead, it:
- Crosses cell membranes using its alternating aromatic-cationic structure
- Accumulates >1000-fold in mitochondria within minutes, driven by membrane potential
- Binds cardiolipin — a phospholipid essential for electron transport chain (ETC) complex stability
- Stabilizes ETC supercomplexes — improving electron transfer efficiency
- Result: More ATP per oxygen consumed, less electron leak, less ROS
This is a structural fix — SS-31 restores the physical architecture of the inner mitochondrial membrane. Think of it as re-seating loose circuit board connections so the system runs efficiently again.
The cardiolipin binding mechanism was directly demonstrated by Birk et al., showing SS-31 interacts selectively with cardiolipin to re-energize ischemic mitochondria (Birk et al., 2013).
For the full SS-31 mechanism breakdown, see our SS-31 Research Guide.
MOTS-c: AMPK Activation (Signaling)
MOTS-c works through cellular signaling — it activates a metabolic master switch:
- Secreted from mitochondria into the cytoplasm (and potentially into circulation as a "mitokine")
- Inhibits the folate-methionine cycle — specifically the de novo purine biosynthesis pathway
- Activates AMPK (AMP-activated protein kinase) — the cell's primary energy sensor
- AMPK translocates to the nucleus — directly regulating gene expression
- Result: Enhanced glucose uptake, improved insulin sensitivity, fatty acid oxidation, exercise-like metabolic effects
This is a signaling cascade — MOTS-c triggers a metabolic program that mimics some effects of exercise. Lee et al. first characterized MOTS-c as a mitochondrial-derived peptide that regulates metabolic homeostasis through AMPK (Lee et al., 2015).
The Key Distinction
|
SS-31 |
MOTS-c |
| Works where? |
Inside mitochondria (inner membrane) |
Outside mitochondria (cytoplasm → nucleus) |
| How? |
Physical stabilization of membrane lipid |
Enzyme inhibition → kinase activation → gene regulation |
| Speed |
Minutes (biophysical binding) |
Hours to days (transcriptional changes) |
| Analogy |
Mechanic fixing the engine |
Software update changing how the car drives |
These mechanisms are completely non-overlapping — SS-31 fixes mitochondrial hardware while MOTS-c reprograms cellular metabolic software.
Research Applications
Where SS-31 Has Stronger Data
SS-31's research strength is in tissues with high mitochondrial demand where structural damage drives disease:
-
Heart failure — Phase II/III clinical trials (TAZPOWER, PROGRESS-HF). SS-31 improved cardiac function by restoring mitochondrial efficiency in energy-starved cardiomyocytes (Thompson et al., 2021)
-
Barth syndrome — A genetic cardiolipin deficiency. SS-31 directly addresses the molecular defect — making it one of the most rationally targeted peptide therapies in development
-
Age-related mitochondrial decline — Siegel et al. showed a single SS-31 injection restored mitochondrial ADP sensitivity in aged mice to near-youthful levels within 1 hour (Siegel et al., 2013)
-
Ischemia-reperfusion injury — Kidney, heart, and brain models show SS-31 protects mitochondria during the critical reperfusion window when ROS generation peaks (Szeto et al., 2011)
-
Skeletal muscle aging — 8 weeks of SS-31 reversed age-related redox stress and improved exercise tolerance in aged mice (Campbell et al., 2019)
Where MOTS-c Has Stronger Data
MOTS-c's research strength is in metabolic regulation and exercise biology:
-
Obesity & metabolic syndrome — MOTS-c treatment prevented age-dependent and high-fat-diet-induced insulin resistance in mice. It improved glucose tolerance and reduced fat accumulation (Lee et al., 2015)
-
Exercise mimetic effects — MOTS-c activates AMPK and downstream metabolic pathways that overlap with exercise signaling. It has been described as an "exercise mimetic" peptide (Lee et al., 2015)
-
Insulin sensitivity — Directly improves glucose uptake through AMPK-mediated GLUT4 translocation, independent of insulin signaling
-
Aging & cellular stress response — MOTS-c translocates to the nucleus during metabolic stress to regulate adaptive gene expression. Reynolds et al. demonstrated this nuclear translocation as a key mechanism linking mitochondrial signaling to nuclear gene regulation (Reynolds et al., 2021)
-
Circulating biomarker — MOTS-c levels decline with age in humans, suggesting a role as a measurable aging biomarker and potential therapeutic target
Head-to-Head Comparison

| Research Area |
SS-31 Evidence |
MOTS-c Evidence |
| Heart failure |
Phase II/III clinical trials |
No direct data |
| Skeletal muscle aging |
Strong preclinical + clinical |
Moderate preclinical |
| Metabolic syndrome / obesity |
Limited |
Strong preclinical |
| Insulin sensitivity |
Indirect (via ATP restoration) |
Direct (via AMPK → GLUT4) |
| Exercise capacity |
Improved in aged mice |
Exercise mimetic in mice |
| Ischemia-reperfusion |
Strong preclinical |
No direct data |
| Neurodegeneration |
Emerging preclinical |
Emerging preclinical |
| Human clinical data |
Yes (Phase II/III) |
No |
Dosing Comparison
| Parameter |
SS-31 |
MOTS-c |
| Clinical dose |
4–40 mg SC daily |
No clinical trials |
| Preclinical dose |
0.1–3 mg/kg SC/IP |
5–15 mg/kg IP |
| Route |
SC (clinical), IV (Phase I), IP (preclinical) |
IP only (preclinical) |
| Frequency |
Once daily (clinical) |
Variable (preclinical) |
| Duration |
4–168 weeks (clinical) |
1–8 weeks (preclinical) |
| Onset |
Mitochondrial effects within 1 hour |
Metabolic effects over hours to days |
For complete SS-31 dosing protocols, see our SS-31 Dosing Guide.
Safety & Clinical Development
|
SS-31 |
MOTS-c |
| Human safety data |
Phase I/II/III trials |
None |
| Serious adverse events |
None reported in clinical trials |
Unknown in humans |
| Common side effects |
Injection site reactions, mild headache |
Unknown in humans |
| FDA status |
Not approved; active clinical development |
Not in clinical development |
| Orphan drug designation |
Yes (Barth syndrome) |
No |
SS-31 has a significant clinical development advantage — it is the only mitochondrial peptide with Phase III trial data. MOTS-c remains entirely preclinical, though its endogenous nature (naturally produced by mitochondria) provides a theoretical safety baseline.
Can They Be Combined?
Because SS-31 and MOTS-c work through completely non-overlapping mechanisms, they represent a logical combination in research:
- SS-31 restores mitochondrial bioenergetics from the inside (structural fix)
- MOTS-c activates metabolic signaling from the outside (regulatory fix)
This is analogous to combining a hardware repair (SS-31 fixing the ETC) with a software update (MOTS-c activating AMPK metabolic programs). No published studies have evaluated this combination, but their mechanistic independence suggests no pharmacological conflict.
Other longevity peptides that could complement both:
- Epitalon — Telomerase activation for cellular longevity (different target: telomeres)
- Humanin — Cytoprotective signaling (different target: anti-apoptotic pathways)
- NAD+ — Coenzyme for cellular energy and DNA repair (different target: sirtuin activation)
For combination protocols, see our peptide stacking guide.
Which Should You Research?
| If your research focuses on... |
Consider |
| Heart failure / cardiac protection |
SS-31 (clinical trial data) |
| Age-related mitochondrial decline |
SS-31 (strongest preclinical data) |
| Metabolic syndrome / insulin resistance |
MOTS-c (direct AMPK mechanism) |
| Exercise biology / performance |
MOTS-c (exercise mimetic data) |
| Ischemia-reperfusion injury |
SS-31 (rapid protection) |
| Obesity / fat metabolism |
MOTS-c (metabolic regulation) |
| Broadest safety data |
SS-31 (Phase II/III trials) |
| Aging biomarker research |
MOTS-c (circulating levels decline with age) |
Frequently Asked Questions
Is SS-31 better than MOTS-c?
Neither is objectively better — they target completely different aspects of mitochondrial biology. SS-31 fixes mitochondrial structure (cardiolipin). MOTS-c activates metabolic signaling (AMPK). The "better" choice depends entirely on the research question.
Can SS-31 and MOTS-c be used together?
No published studies have evaluated this combination, but their mechanisms are entirely non-overlapping (cardiolipin binding vs. AMPK activation), suggesting no pharmacological conflict. This remains a theoretical combination.
Which has more human data?
SS-31 by a wide margin. It has Phase I, II, and III clinical trials with published safety and efficacy data. MOTS-c has no human clinical trials — all data is preclinical (animal models and cell culture).
Are there other mitochondrial peptides?
Yes. Humanin is another mitochondrial DNA-encoded peptide with anti-apoptotic and neuroprotective properties. Epitalon targets telomerase rather than mitochondria directly but is often studied in the same longevity context. See our longevity peptides category for the full list.
References
-
Zhao K, Zhao GM, Wu D, et al. "Cell-permeable peptide antioxidants targeted to inner mitochondrial membrane." J Biol Chem. 2004;279(33):34682-90. PubMed
-
Birk AV, Liu S, Soong Y, et al. "The mitochondrial-targeted compound SS-31 re-energizes ischemic mitochondria by interacting with cardiolipin." J Am Soc Nephrol. 2013;24(8):1250-61. PubMed
-
Siegel MP, Kruse SE, Percival JM, et al. "Mitochondrial-targeted peptide rapidly improves mitochondrial energetics and skeletal muscle performance in aged mice." Aging Cell. 2013;12(5):763-71. PubMed
-
Szeto HH. "First-in-class cardiolipin-protective compound as a therapeutic agent to restore mitochondrial bioenergetics." Br J Pharmacol. 2014;171(8):2029-50. PubMed
-
Thompson R, Hornby B, Manuel R, et al. "A phase 2/3 randomized clinical trial followed by an open-label extension to evaluate the effectiveness of elamipretide in Barth syndrome." Genet Med. 2021;23(12):2364-72. PubMed
This article is for educational and research purposes only. It is not medical advice.


