Best Peptides for Energy and Fatigue (2026)
Fatigue is not a single problem -- it is a symptom with at least four distinct biological mechanisms: mitochondrial dysfunction (your cells cannot produce enough ATP), hormonal decline (growth hormone, thyroid, testosterone deficits), sleep architecture disruption (insufficient deep sleep and REM), and metabolic inflexibility (impaired ability to switch between fuel sources). Treating "low energy" without identifying the underlying mechanism is why most supplements fail.
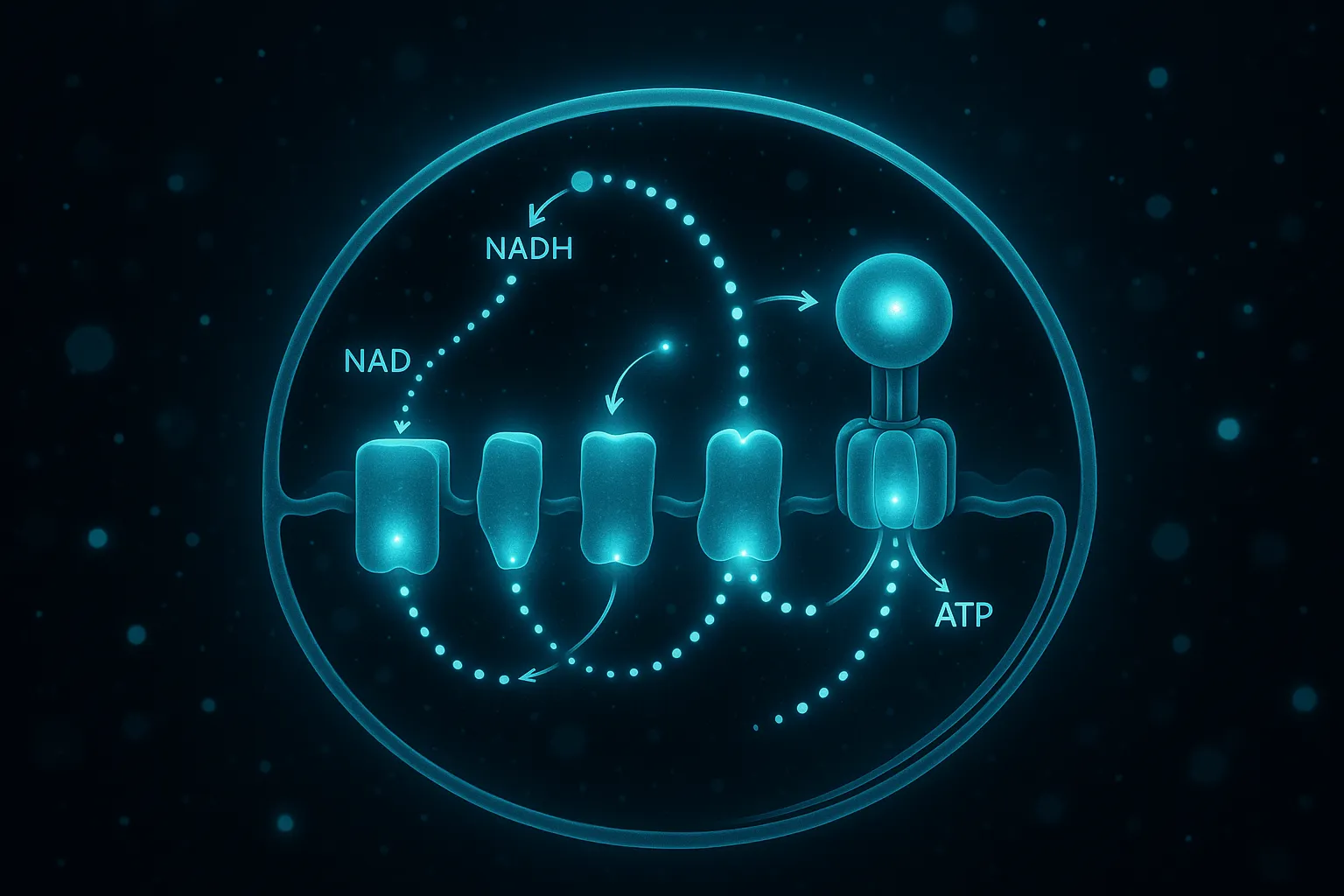
Peptide therapy offers targeted interventions for each of these mechanisms. Mitochondrial-targeted peptides like SS-31 and MOTS-c directly enhance the electron transport chain and cellular energy metabolism. Growth hormone secretagogues restore the anabolic and sleep-promoting effects of GH that decline with age. NAD+ precursors replenish the central cofactor that mitochondria need to produce ATP. And each approach works through a different enough pathway that they can be meaningfully combined.
This article ranks 5 peptides and peptide-related compounds for energy and fatigue by their mechanism, evidence strength, and the specific type of fatigue they address. Each section links to the full peptide page for detailed protocols.
Quick Comparison Table
| Peptide | Mechanism | Fatigue Type Targeted | Evidence Level | Route |
|---|---|---|---|---|
| MOTS-c | AMPK activation + metabolic regulation | Metabolic/insulin-related fatigue | Preclinical + early human | Injectable |
| SS-31 (Elamipretide) | Mitochondrial inner membrane stabilization | Age-related energy decline | Phase 2/3 (cardiac) + preclinical | Injectable |
| NAD+ Precursors (NR/NMN) | NAD+ cofactor restoration | Age-related NAD+ depletion | Multiple RCTs in humans | Oral |
| CJC-1295/Ipamorelin | GH secretagogue (GHRH + ghrelin mimetic) | Hormonal/recovery fatigue | Phase 2 (CJC-1295) + preclinical | Injectable |
| MK-677 (Ibutamoren) | Oral GH secretagogue (ghrelin mimetic) | Sleep-related fatigue | Phase 2/3 in humans | Oral |
1. MOTS-c -- Mitochondrial Exercise Mimetic
MOTS-c is a 16-amino-acid peptide encoded within the mitochondrial 12S rRNA gene that functions as an endogenous metabolic regulator. It represents the most direct pharmacological approach to the exercise-mimetic concept: enhancing cellular energy metabolism through the same AMPK-mediated pathways that physical activity activates, without requiring the physical activity itself.
The foundational study demonstrated that MOTS-c prevents high-fat-diet-induced obesity, reverses established insulin resistance, and enhances glucose metabolism in skeletal muscle through activation of the AMPK-AICAR pathway [1]. MOTS-c targets the same pathway that metformin activates (AMPK), but does so as a naturally occurring mitochondrial signal peptide rather than a synthetic pharmaceutical.
For energy and fatigue specifically, the exercise capacity data is compelling. MOTS-c treatment in both young and aged mice significantly enhanced physical performance, with the most striking results in older animals. Late-life MOTS-c treatment increased physical capacity and healthspan in aged mice, with adaptations similar to those seen in physically active animals: improved antioxidant capacity, better insulin sensitivity, and enhanced metabolic flexibility [2].
The human connection is established through exercise physiology research. Endogenous MOTS-c levels rise in skeletal muscle and circulation after physical activity, and this exercise-induced MOTS-c expression correlates with the metabolic benefits of exercise. This suggests MOTS-c plays a natural role in translating physical activity into metabolic improvement -- and that supplementation may partially replicate these benefits when exercise capacity is limited by fatigue, injury, or age.
The primary limitation is that large-scale human efficacy trials for energy and fatigue outcomes have not been completed. The biological rationale is strong, the animal data is consistent, and the endogenous role in exercise physiology is established -- but the clinical proof gap remains.
MOTS-c is administered via subcutaneous injection. For current evidence and protocols, see the MOTS-c page.
Top MOTS-C Vendors
Ranked by price, COA availability, and reputation
2. SS-31 (Elamipretide) -- Mitochondrial Membrane Stabilizer
SS-31 (also known as elamipretide) is a tetrapeptide that targets the inner mitochondrial membrane with remarkable specificity. It binds to cardiolipin, a phospholipid unique to the mitochondrial inner membrane, and stabilizes the electron transport chain complexes that produce ATP. This is the most direct intervention point for cellular energy production available in peptide form.
The energy-relevant evidence is substantial. In aged mice, 8 weeks of SS-31 treatment reversed age-related mitochondrial dysfunction, restored redox homeostasis, and improved exercise tolerance -- without increasing mitochondrial content [3]. This means SS-31 does not make more mitochondria; it makes existing mitochondria work better. The improvement in exercise capacity was achieved by normalizing proton leak (a major source of mitochondrial inefficiency in aging) and reducing mitochondrial reactive oxygen species production.
The cardiac data extends this principle to the most energy-demanding organ. Late-life SS-31 treatment substantially reversed cardiac dysfunction in old mice, normalizing the increase in proton leak and reducing mitochondrial ROS in cardiomyocytes [4]. This cardiac improvement has direct implications for exercise intolerance and fatigue -- cardiac output limitations are a primary contributor to exercise capacity decline with aging.
SS-31 also improved ADP sensitivity in aged mitochondria by increasing uptake through the adenine nucleotide translocator (ANT) [5]. ADP sensitivity is a critical determinant of how quickly mitochondria can ramp up ATP production in response to energy demand -- poor ADP sensitivity means sluggish energy responses during physical or mental exertion, which manifests as fatigue.
The clinical development of SS-31 has focused on mitochondrial diseases (Barth syndrome) and heart failure, where it has reached phase 2/3 trials. For age-related fatigue specifically, the preclinical evidence is the strongest available for any mitochondrial-targeted peptide, but human trials for this indication are needed.
SS-31 is administered via subcutaneous injection or intravenous infusion in clinical settings.
Top SS-31 (Elamipretide) Vendors
Ranked by price, COA availability, and reputation