You have tried the willpower approach. You have tried the supplement approach — fiber capsules, green tea extract, garcinia cambogia, the latest "metabolism booster" from a sponsored Instagram post. Maybe something worked for a week or two. Then your appetite came back, stronger than before, and the weight followed.
This is not a willpower failure. It is a biology problem. Your brain's appetite center — the hypothalamus — is one of the most persistent regulatory systems in the body. It evolved to prevent starvation, and it treats caloric restriction as a threat. When you cut calories, your hypothalamus fights back: it increases hunger hormones (ghrelin), decreases satiety hormones (leptin sensitivity), reduces metabolic rate, and makes food more rewarding neurologically. Most supplements cannot overcome this system because they do not reach the receptors that control it.
Understanding why conventional appetite suppressants fail is the first step toward finding approaches that actually work long-term.
Conventional Approaches: What Helps and What Falls Short
High-protein diets genuinely suppress appetite — and the evidence is strong. Protein triggers greater satiety per calorie than carbohydrates or fat, and it has a higher thermic effect (your body burns more calories digesting it). If you are eating less than 0.7g of protein per pound of body weight, increasing protein intake is the single most impactful dietary change for appetite management. It is free, has no side effects, and works.
Fiber supplementation (glucomannan, psyllium husk) provides modest appetite reduction by expanding in the stomach and slowing gastric emptying. The effects are real but small — typically resulting in 1-3 pounds of additional weight loss over several months in controlled studies. Useful as a complement, not as a primary strategy.
Green tea extract and caffeine mildly increase metabolic rate and can temporarily suppress appetite. Tolerance builds quickly, and the effect size is small — roughly 50-100 extra calories burned per day, which is equivalent to half an apple. These compounds are not meaningless, but they are not moving the needle for anyone with significant appetite issues.
5-HTP targets serotonin synthesis, and serotonin does regulate satiety. Some studies show modest appetite reduction. However, the effect is inconsistent, and long-term serotonin manipulation without medical supervision carries risks (serotonin syndrome when combined with SSRIs, for example).
Prescription appetite suppressants like phentermine work by stimulating norepinephrine release — essentially, they are amphetamine-adjacent stimulants. They are effective short-term (3-6 months) but carry cardiovascular side effects, tolerance development, and rebound hunger when discontinued. The FDA limits phentermine to short-term use for this reason.
Bariatric surgery is effective and well-studied — patients lose 25-35% of body weight and keep most of it off long-term. But it is irreversible, carries surgical risks, requires permanent dietary changes, and is typically reserved for people with BMI over 40 (or 35 with comorbidities).
The gap: the brain's appetite regulation system is extraordinarily persistent. Most over-the-counter supplements do not interact with the specific receptors (GLP-1, GIP, glucagon) that govern hunger signaling in the hypothalamus. Prescription stimulants reach those circuits but through blunt mechanisms with tolerance and rebound issues. The biological question is: can you target the appetite signaling pathways precisely, with sustained effect, and without the limitations of stimulant-based approaches?

The Research Frontier: Targeting Hunger at the Receptor Level
Over the past decade, a class of compounds called incretin-based peptides has fundamentally changed the landscape of appetite management. These compounds mimic natural gut hormones — specifically GLP-1 (glucagon-like peptide-1) and GIP (glucose-dependent insulinotropic polypeptide) — that your body already uses to regulate hunger, satiety, and blood sugar.
Peptides are short chains of amino acids that act as signaling molecules. Incretin peptides bind to the same receptors that your natural gut hormones use, but with much longer duration of action. Where your natural GLP-1 is degraded within minutes, synthetic GLP-1 agonists maintain receptor activation for days — providing sustained appetite suppression that natural hormones cannot.
The clinical trial data for these compounds is not preliminary. It represents the largest body of obesity pharmacotherapy evidence ever generated — tens of thousands of participants across dozens of randomized controlled trials, published in the highest-impact medical journals.
For a foundational understanding of how peptides work as signaling molecules, see What Are Peptides?.
How Incretin Peptides Suppress Appetite Differently
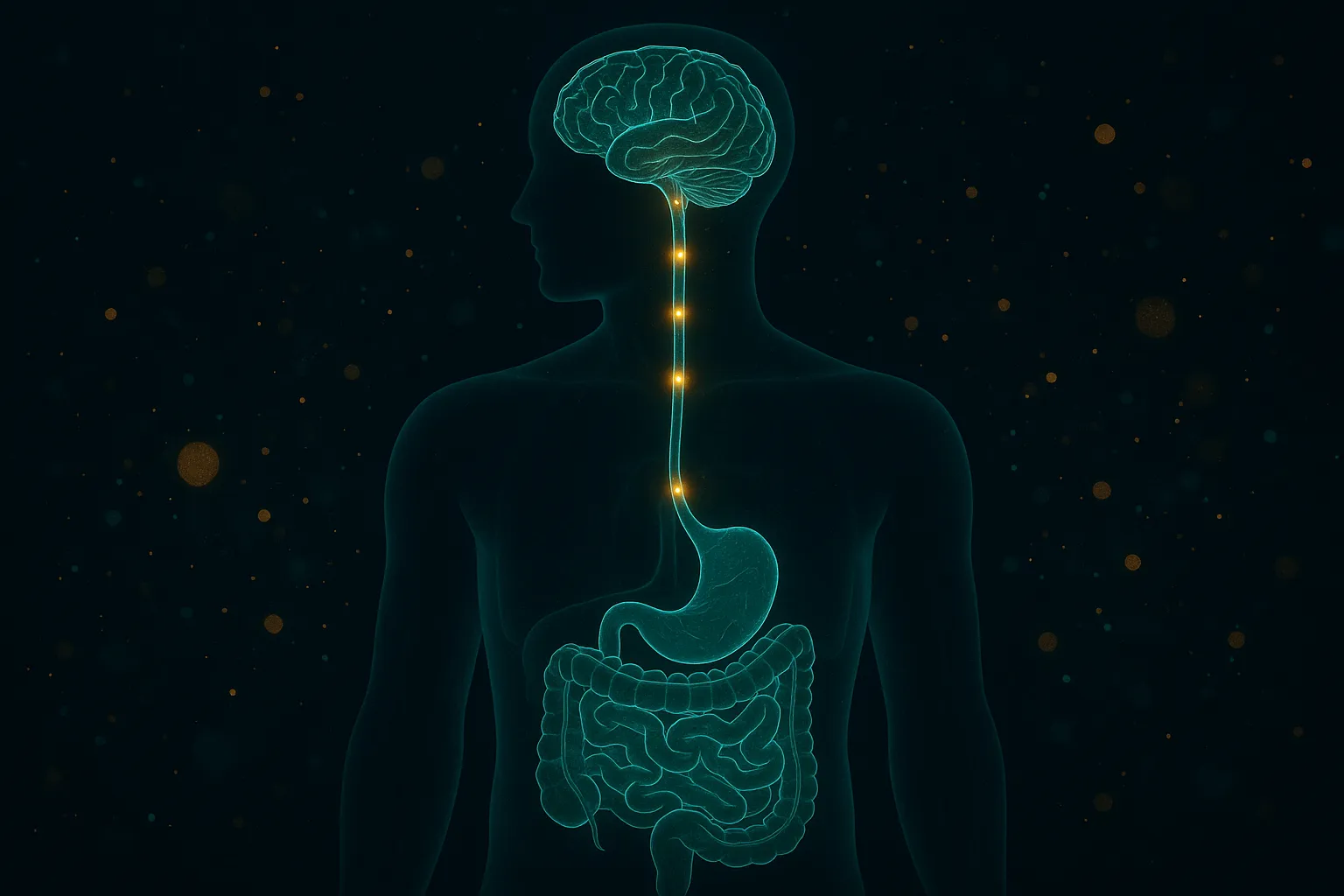
Traditional appetite suppressants work in the brain — stimulating norepinephrine or serotonin to override hunger signals. Incretin peptides work at the source: the gut-brain axis.
When you eat, your intestines release GLP-1 and GIP to signal satiety to the brain. These hormones tell the hypothalamus "you have eaten enough," slow gastric emptying (so food stays in your stomach longer), and improve insulin sensitivity (so nutrients are processed more efficiently). In people struggling with appetite regulation, this signaling system is often blunted — the signals are too weak or too short-lived to overcome the hypothalamus's drive to eat.
Synthetic GLP-1 agonists amplify these signals. They bind the same receptors but resist enzymatic degradation, maintaining satiety signaling for days rather than minutes. The result is not stimulant-driven appetite masking — it is a restoration (and amplification) of the natural "I am full" pathway. People on these compounds consistently describe the experience as simply not thinking about food, rather than fighting cravings.
The Key Compounds for Appetite Management
Semaglutide — The Most-Studied Weight Loss Peptide
Semaglutide is a GLP-1 receptor agonist with the most extensive clinical evidence of any weight loss compound. The STEP 1 trial — a 68-week randomized controlled study of 1,961 adults — demonstrated a mean weight loss of 14.9% with semaglutide 2.4 mg weekly versus 2.4% with placebo (PMID:33567185). That is roughly 33 pounds for a 220-pound person.
Semaglutide works by activating GLP-1 receptors in the gut and brain, reducing appetite, slowing gastric emptying, and improving glycemic control. It is administered as a once-weekly injection and is available through prescription from healthcare providers and telehealth platforms.
The evidence base is massive — multiple phase 3 trials, long-term follow-up data, and cardiovascular outcome studies. For anyone evaluating appetite management options, semaglutide is the benchmark against which everything else is measured.
Top Semaglutide Vendors
Ranked by price, COA availability, and reputation
Tirzepatide — Dual Receptor Agonist
Tirzepatide activates two receptors instead of one: both GLP-1 and GIP. This dual mechanism produced the highest weight loss numbers of any approved compound. The SURMOUNT-1 trial showed that participants on 15 mg tirzepatide lost an average of 22.5% of body weight over 72 weeks, with 57% achieving 20% or greater weight reduction (PMID:35658024).
The GIP receptor activation adds a distinct metabolic benefit beyond what GLP-1 alone provides. GIP influences fat tissue metabolism, insulin secretion, and energy balance through pathways that complement GLP-1's appetite suppression. The result is greater weight loss with a similar side effect profile.
Tirzepatide is a once-weekly injection, available by prescription. It represents the current efficacy frontier for approved appetite management compounds.
Top Tirzepatide Vendors
Ranked by price, COA availability, and reputation
Retatrutide — Triple Receptor Agonist (Investigational)
Retatrutide targets three receptors: GLP-1, GIP, and glucagon. The addition of glucagon receptor activation drives direct fat oxidation — your body burns stored fat as fuel rather than just reducing caloric intake through appetite suppression.
Phase 2 trial results were striking: participants on the highest dose (12 mg) lost 24.2% of body weight at 48 weeks, and the weight loss curve had not yet plateaued (PMID:37366315). This is the highest percentage weight loss reported for any anti-obesity compound in clinical trials to date.
Retatrutide is still in clinical development — phase 3 trials are ongoing. It is not yet commercially available outside research contexts. Phase 2 results are promising but historically moderate by 2-5 percentage points in larger phase 3 populations. Still, the triple-receptor approach represents where appetite science is heading.
Top Retatrutide Vendors
Ranked by price, COA availability, and reputation
For a full comparison of all fat loss peptides — including non-incretin options like tesamorelin and tesofensine — see Best Peptides for Fat Loss.