Sermorelin is a synthetic version of the first 29 amino acids of human growth hormone-releasing hormone (GHRH). It was formerly FDA-approved for GH deficiency diagnosis, giving it a unique history of clinical safety data compared to most research peptides.
Sermorelin was withdrawn from the market for commercial reasons, not safety concerns. It is currently available only as a research chemical. This is not medical advice.
Quick Reference: Standard Protocol
| Parameter |
Detail |
| Vial |
2 mg |
| BAC Water |
2 mL |
| Concentration |
1,000 mcg/mL |
| Dose |
200-300 mcg (20-30 units on insulin syringe) |
| Route |
Subcutaneous |
| Timing |
Before bed, empty stomach |
| Frequency |
5 days on, 2 days off |
| Cycle |
8 weeks on, 8 weeks off |
| Storage |
Refrigerate, use within 14-28 days |
Key detail: Bedtime dosing aligns with natural GH release during deep sleep. Empty stomach required — wait 2-3 hours after eating, avoid food for 1 hour post-injection.
For the full Sermorelin profile and vendor pricing, see our Sermorelin peptide page.
Cycling Details
Start at 200 mcg daily for weeks 1-2 to assess tolerance, then increase to 250-300 mcg if response warrants. The 5-on/2-off schedule prevents GHRH receptor desensitization — the 2-day break allows receptors to reset.
Sermorelin's short half-life (8-12 minutes) mimics natural GHRH pulses, preserving pulsatile GH patterns rather than creating sustained elevation. This is actually beneficial — it means normal feedback mechanisms remain intact.
Standard cycle is 8 weeks on, 8 weeks off. Some extend to 12-16 weeks with periodic breaks. Competition prep protocols may run 12-16 weeks leading up to an event.
Routes of Administration
Subcutaneous injection is the standard route. Inject before bed at the same time nightly for consistency. Abdomen, thigh — standard SC sites. Volume is 0.2-0.3 mL with insulin syringe (29-31 gauge).
Intramuscular is an alternative but less common — no significant difference in effectiveness vs. SC.
Not oral — Sermorelin is a peptide destroyed by stomach acid (unlike MK-677, which is orally bioavailable).
Reconstitution Quick Reference
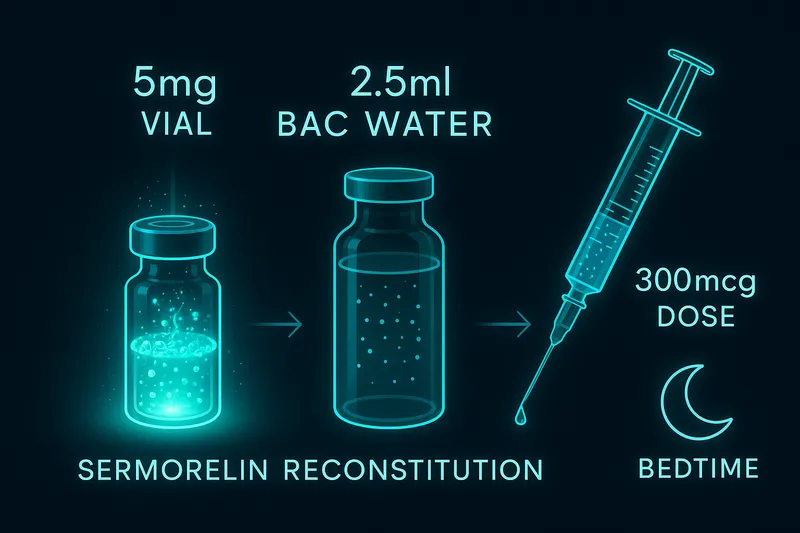
| Vial |
BAC Water |
Concentration |
200 mcg |
250 mcg |
300 mcg |
| 2 mg |
2 mL |
1,000 mcg/mL |
20 units |
25 units |
30 units |
| 5 mg |
2 mL |
2,500 mcg/mL |
8 units |
10 units |
12 units |
Math: 2 mg / 2 mL = 1,000 mcg/mL. For 200 mcg: 200 / 1,000 = 0.2 mL = 20 units.
Swirl gently — do not shake. Refrigerate immediately, use within 14-28 days (more fragile than some peptides). Protect from light. Powder can be stored at -20 C before reconstitution.
Where These Numbers Come From
Sermorelin has a unique advantage — genuine human clinical data from its FDA-approved era.
FDA-approved diagnostic use: 1 mcg/kg IV for GH stimulation testing, establishing safety across age groups (Prakash & Bhatt, 2020).
Community dose rationale: The 200-300 mcg SC range accounts for lower SC bioavailability vs. IV, and targets sustained GH elevation rather than acute diagnostic testing. The original GHRH characterization by Thorner et al. established the peptide's pharmacology (Thorner et al., 1985).
Pharmacokinetics: Half-life of 8-12 minutes. Peak GH effect 15-30 minutes post-injection. GH elevation lasts 1-2 hours. Rapidly cleared by kidney and liver. This short half-life preserves natural pulsatile patterns — a key advantage over long-acting analogs like CJC-1295.
Stacking Protocols
| Stack |
Purpose |
Protocol |
| Sermorelin + Ipamorelin |
Classic GHRH + GHRP — synergistic via different receptors |
Both 200-300 mcg SC, before bed |
| Sermorelin + GHRP-2 |
Traditional anti-aging clinic combination |
Sermorelin 250 mcg + GHRP-2 200-300 mcg, 5on/2off |
Do not stack with CJC-1295 — both are GHRH analogs competing for the same receptor. Choose one based on preference (sermorelin = natural structure/short half-life/daily; CJC-1295 = modified/long half-life/less frequent). Can mix compatible peptides in the same syringe.
Side Effects & Safety
- Injection site reactions — mild redness or swelling, resolves quickly (~5% of users)
- Flushing — brief facial warmth
- Dizziness — mild and transient (uncommon)
- Headache — uncommon, typically mild
- Excellent safety profile from years of FDA-approved clinical use
- No significant drug interactions identified during clinical era
- Contraindications: Active cancer (theoretical concern with growth-promoting compounds), pregnancy/breastfeeding, severe kidney or liver disease
Frequently Asked Questions
What is the standard Sermorelin dose?
200-300 mcg subcutaneously before bed on an empty stomach, 5 days on / 2 days off, 8 weeks on / 8 weeks off.
Why was Sermorelin discontinued?
Withdrawn for commercial reasons (limited market viability for the diagnostic indication), not safety concerns.
Why the 5 days on, 2 days off schedule?
Prevents GHRH receptor desensitization. The 2-day break allows receptors to reset and preserves natural pulsatile GH patterns.
Can Sermorelin be taken with food?
No — empty stomach required. Wait 2-3 hours after eating, avoid food for 1 hour post-injection.
What's the difference between Sermorelin and CJC-1295?
Sermorelin has a short half-life (8-12 minutes) mimicking natural GHRH pulses, requiring daily dosing. CJC-1295 is modified for extended duration (8+ days) with less frequent dosing. Sermorelin better preserves natural rhythms.
Does Sermorelin need to be refrigerated?
Yes — both powder and solution require refrigeration. Use within 14-28 days. Do not freeze reconstituted solution.
References
| Citation |
Topic |
PMID |
| Prakash & Bhatt, Cureus (2020) |
Sermorelin clinical review, diagnostic use, safety |
32123586 |
| Walker, Mol Cell Endocrinol (2006) |
GHRH mechanism, receptor binding, clinical applications |
16880825 |
| Thorner et al., J Clin Endocrinol Metab (1985) |
Original Sermorelin (GHRH 1-29) characterization |
4019717 |
| Gelato et al., J Clin Endocrinol Metab (1984) |
GHRH dose-response, pharmacokinetics |
6480785 |
For educational and research purposes only. This is not medical advice. Sermorelin is not currently FDA-approved and is available only as a research chemical.

