People throw around GHRH, GHRP, and secretagogue like they're interchangeable. They're not.
They all can increase growth hormone (GH) signaling in some context, but they do it through different receptors, different intracellular pathways, and often different side-effect profiles.
Quick Definitions
GHRH (Growth Hormone-Releasing Hormone)
A natural hypothalamic hormone that signals the pituitary to release GH. GHRH is a 44-amino acid peptide produced in the arcuate nucleus of the hypothalamus and released into the hypophyseal portal circulation to reach anterior pituitary somatotrophs.
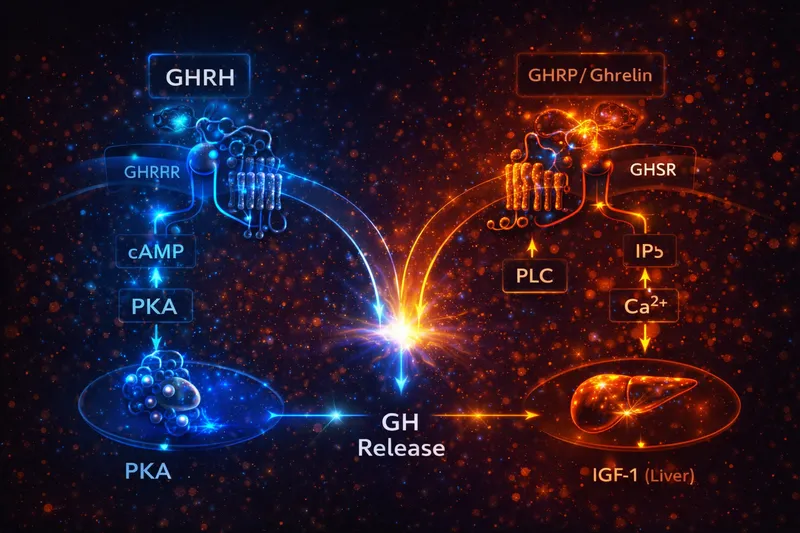
- Receptor: GHRH receptor (GHRHR) on pituitary somatotrophs - a class B G protein-coupled receptor
- Main signaling: Gs → adenylyl cyclase → cAMP → PKA (with calcium involvement downstream via L-type voltage-gated calcium channels)
- Net effect: Promotes GH synthesis and release in a physiologic "pulse-friendly" way, still modulated by somatostatin, sleep, nutrition, and IGF-1 feedback
The GHRHR couples primarily to Gαs, activating adenylyl cyclase to increase intracellular cAMP. This activates protein kinase A (PKA), which phosphorylates CREB and other transcription factors to drive GH gene expression. PKA also facilitates GH vesicle exocytosis through calcium channel modulation (PubMed: Regulation of the pituitary somatotroph cell by GHRH and its receptor).
Examples include Sermorelin, CJC-1295, and Tesamorelin. These are GHRH analogs that activate the same receptor as natural GHRH but often with modified pharmacokinetics (e.g., CJC-1295 with DAC has extended half-life via albumin binding).
GHRP (Growth Hormone-Releasing Peptide)
Synthetic peptide class that mimics the GH-stimulating effects of the ghrelin system. GHRPs were discovered before ghrelin itself - researchers found these synthetic peptides released GH through an unknown receptor, which was later identified as the ghrelin receptor (GHSR) when ghrelin was discovered in 1999.
- Receptor: Growth hormone secretagogue receptor type 1a (GHSR-1a), also known as the ghrelin receptor
- Main signaling: Gq/11 → phospholipase C (PLC) → IP3 → intracellular Ca²⁺ release from ER stores, plus direct effects on hypothalamic neurons
- Net effect: Stimulates GH release through both pituitary and hypothalamic actions; often has stronger orexigenic (appetite-stimulating) effects than GHRH analogs
GHSR-1a is a constitutively active receptor, meaning it has significant basal signaling even without ligand binding. This constitutive activity is important for body weight regulation. When activated by ghrelin or GHRPs, the receptor couples primarily to Gq/11, activating PLC-β to generate IP3 and diacylglycerol (DAG). IP3 triggers calcium release from the endoplasmic reticulum, while DAG activates protein kinase C (PubMed: Ghrelin receptor signaling in health and disease).
Examples include Ipamorelin, GHRP-2, GHRP-6, and Hexarelin. These peptides vary in their selectivity and side-effect profiles - for instance, Ipamorelin is notable for minimal effects on cortisol and prolactin compared to GHRP-6.
"Secretagogues"
An umbrella term meaning "something that stimulates secretion." In the GH world, "secretagogues" often refers to:
- GHRH analogs (GHRH-like): Work through GHRHR
- Ghrelin mimetics / GHSR agonists (GHRP-like): Work through GHSR-1a, includes peptides and non-peptides
So GHRH and GHRP are both "secretagogues," but they operate through completely different mechanisms. This distinction matters for understanding their effects, side-effect profiles, and why they're often combined.
The GH Axis: Understanding Pulsatile Secretion
GH secretion is not continuous - it occurs in discrete pulses, with the largest pulses occurring during deep sleep. This pulsatility is essential for GH's physiological effects; continuous GH exposure leads to receptor desensitization and different downstream signaling patterns than pulsatile exposure.
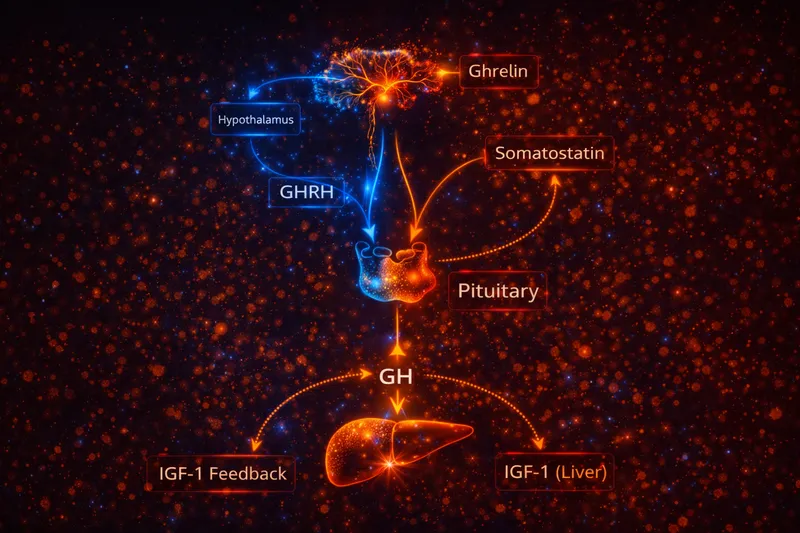
GH secretion is regulated by three main inputs:
- GHRH (stimulatory) - Released from the arcuate nucleus, promotes GH synthesis and release
- Somatostatin (inhibitory) - Released from the periventricular nucleus, inhibits GH release but not synthesis
- Ghrelin signaling (stimulatory via GHSR) - Released primarily from the stomach, amplifies GH pulses and links nutritional status to GH secretion
These inputs integrate at the pituitary and hypothalamus to produce pulsatile GH release. The current model suggests that GH pulses occur when GHRH release coincides with somatostatin withdrawal (the "somatostatin withdrawal" hypothesis). Ghrelin amplifies these pulses, particularly in states of negative energy balance (PubMed: Physiological role of somatostatin on growth hormone regulation).
GH then drives many downstream effects via IGF-1 (insulin-like growth factor 1), which is produced primarily by the liver but also locally in target tissues. IGF-1 provides negative feedback to the hypothalamus and pituitary, completing the regulatory loop (PubMed: Interrelationship between ghrelin and somatostatin/GHRH).
Why pulsatility matters: GH biology isn't just "more GH = more effect." The pattern of GH exposure determines tissue responses. For example, the sexually dimorphic pattern of GH secretion (more pulsatile in males, more continuous in females) drives different liver gene expression profiles. Therapeutic approaches that preserve or enhance natural pulsatility may have advantages over those that simply elevate mean GH levels.
Mechanism Deep-Dive: GHRH vs GHRP
1) Receptor Targets and Sites of Action
GHRH → GHRHR (Primarily Pituitary)
GHRH is the canonical "turn on GH" signal from the hypothalamus to the pituitary. The GHRH receptor is expressed almost exclusively on pituitary somatotrophs, making GHRH action relatively tissue-specific.
Key features of GHRHR signaling:
- GHRHR activation increases cAMP/PKA signaling in somatotrophs
- This drives both immediate GH release (vesicle exocytosis) and longer-term GH synthesis (gene transcription via CREB/Pit-1)
- The pathway also opens L-type calcium channels, contributing to vesicle fusion and release
Practical implication: GHRH analogs provide "pituitary-direct" stimulation that remains under normal physiological regulation. Somatostatin can still inhibit release, and IGF-1 negative feedback remains intact. This makes GHRH analogs relatively "physiologic" in their action (PubMed: GHRH receptor and its signaling).
GHRP / Ghrelin Mimetics → GHSR-1a (Pituitary + Hypothalamus + Peripheral)
GHSR-1a has a broader expression pattern than GHRHR. It's found on:
- Pituitary somatotrophs (direct GH release)
- Hypothalamic neurons in the arcuate nucleus (modulates GHRH/somatostatin neurons)
- Vagal afferents and brainstem nuclei (appetite/reward signaling)
- Other peripheral tissues
This broader expression pattern means GHRPs have effects beyond just GH release:
- Appetite stimulation - GHSR activation in the hypothalamus and brainstem drives hunger signaling
- Reward pathway modulation - GHSR is expressed in dopaminergic circuits
- Gastric motility effects - Ghrelin/GHRPs can promote gastric emptying
Practical implication: You commonly see appetite-related effects with GHRPs because ghrelin biology is built to connect "energy intake" with endocrine output. GHRP-6 is particularly notable for appetite stimulation, while Ipamorelin appears to have less effect on appetite despite activating the same receptor (PubMed: Ghrelin receptor signaling).
2) Intracellular Signaling: cAMP/PKA vs PLC/Ca²⁺
The fundamental difference in intracellular signaling explains many of the distinct characteristics of these peptide classes.
GHRH → GHRHR → Gαs → Adenylyl Cyclase → ↑cAMP → PKA activation
↳ CREB phosphorylation → GH gene transcription
↳ L-type Ca²⁺ channel opening → GH vesicle exocytosis
This follows the classic Gs-coupled receptor paradigm:
- Ligand binding activates Gαs
- Gαs stimulates adenylyl cyclase
- cAMP accumulates and activates PKA
- PKA phosphorylates multiple targets including CREB, ion channels, and exocytotic machinery
The cAMP/PKA pathway is well-suited for sustained, transcriptionally-supported GH release. PKA-mediated CREB phosphorylation works with the pituitary-specific transcription factor Pit-1 to maintain GH gene expression (PubMed: CREB and CBP regulation of GH promoter).
GHRP/ghrelin → GHSR-1a → Gαq/11 → PLC-β → IP3 + DAG
↳ IP3 → Ca²⁺ release from ER → GH vesicle exocytosis
↳ DAG → PKC activation → various downstream effects
The Gq/PLC/calcium pathway produces a more rapid, "burst-like" response:
- Ligand binding activates Gαq/11
- Gαq activates phospholipase C-β (PLC-β)
- PLC-β cleaves PIP2 into IP3 and diacylglycerol (DAG)
- IP3 triggers calcium release from intracellular stores
- The calcium spike triggers rapid vesicle exocytosis
GHSR-1a can also couple to other G proteins (Gαi/o, Gα12/13) and recruit β-arrestins, leading to additional signaling outputs including ERK/MAPK activation. This "biased signaling" may explain why different GHSR agonists have somewhat different effect profiles despite binding the same receptor (PubMed: Structural basis of human ghrelin receptor signaling).
Key differences in signaling character:
- GHRH/cAMP pathway = Signal amplification, sustained response, transcriptional support
- GHRP/calcium pathway = Rapid response, burst-like release, integration with energy-sensing circuits
3) Why People "Stack" Them: The Synergy Rationale
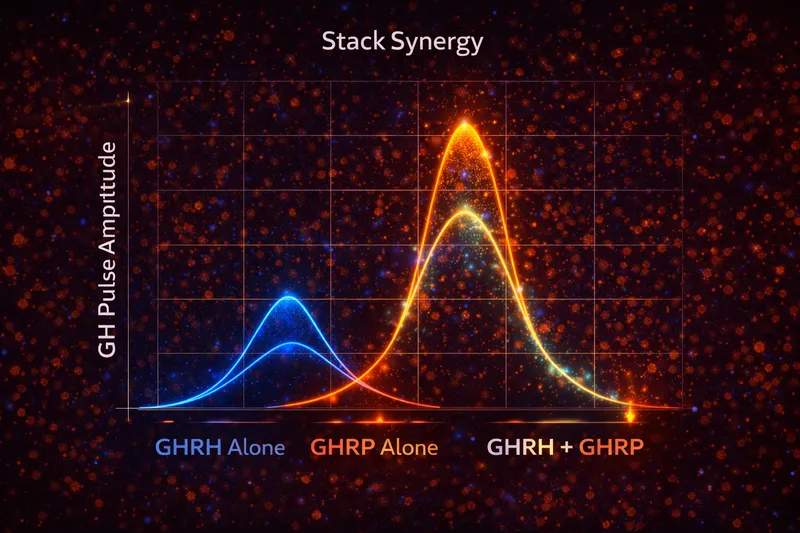
Early research demonstrated that combining GHRH with GHRP produces GH release that exceeds the sum of individual responses - true pharmacological synergy. This was observed before the GHSR was even identified (PubMed: Growth hormone-releasing peptide stimulates GH release synergistically with GHRH).
The mechanistic basis for synergy:
-
Convergent but distinct pathways - GHRH (cAMP) and GHRP (calcium) activate different second messenger systems that converge on GH release. The calcium signal from GHRP can potentiate the cAMP-driven response from GHRH.
-
Dual-site action of GHRPs - GHRPs act at both pituitary and hypothalamus. At the hypothalamic level, they may enhance GHRH release and/or suppress somatostatin, creating a more permissive environment for the direct pituitary effects of administered GHRH.
-
Amplification of physiological pulses - The combination may better mimic the natural situation where endogenous ghrelin rises during fasting and amplifies GHRH-driven GH pulses.
This is the scientific basis for combinations like CJC-1295 + Ipamorelin - the GHRH analog activates GHRHR (cAMP pathway) while the GHRP activates GHSR (calcium pathway). For more on combining peptides, see our Peptide Stacking Guide. The result is enhanced GH pulsatility that neither agent achieves alone (PubMed: Growth hormone-releasing peptides review).
4) Differences in Side-Effect Profiles
The distinct mechanisms also explain different side-effect patterns:
GHRH Analogs:
- Generally well-tolerated
- Minimal appetite effects
- Minimal effects on cortisol, prolactin
- Main concerns: injection site reactions, potential for antibody development with some analogs
GHRPs:
- Appetite stimulation (variable by compound - GHRP-6 > GHRP-2 > Ipamorelin)
- Some GHRPs increase cortisol and prolactin (Hexarelin, GHRP-6 > Ipamorelin)
- Water retention in some users
- The appetite effect can be therapeutic (in cachexia) or unwanted (in obesity research)
Ipamorelin is often preferred among GHRPs because it appears to be more selective for GH release with less effect on cortisol, prolactin, and appetite compared to earlier GHRPs (PubMed: Growth hormone-releasing peptides clinical and basic aspects).
Where MK-677 Fits

Some popular "GH secretagogues" are not peptides. MK-677 (ibutamoren) is an oral, non-peptide ghrelin mimetic / GHSR agonist that has been studied extensively in humans.
Key characteristics of MK-677:
- Oral bioavailability - Unlike peptide GHRPs, MK-677 can be taken orally
- Long half-life - ~24 hours, allowing once-daily dosing
- Mechanism - GHSR-1a agonist, functionally equivalent to GHRPs
- Effects - Increases GH pulsatility, raises IGF-1, stimulates appetite
Clinical studies have shown that MK-677 increases GH secretion and IGF-1 levels in various populations including healthy elderly, GH-deficient adults, and obese subjects. In the elderly, MK-677 restored IGF-1 levels to those of younger adults without major safety concerns in studies up to 2 years (PubMed: MK-677 in healthy elderly).
So MK-677 is a "secretagogue" but it's functionally in the GHRP/ghrelin bucket (GHSR-driven), not the GHRH bucket. This explains why it shares the appetite-stimulating properties common to ghrelin receptor agonists. Like GHRPs, it can be combined with GHRH analogs for potentially synergistic effects (PubMed: Growth hormone-releasing peptides and their analogs).
Summary Table
| Feature |
GHRH Analogs |
GHRPs / Ghrelin Mimetics |
| Receptor |
GHRHR |
GHSR-1a |
| G-protein |
Gαs |
Gαq/11 (primary) |
| Second messenger |
cAMP → PKA |
IP3 → Ca²⁺, DAG → PKC |
| Primary site |
Pituitary (specific) |
Pituitary + Hypothalamus + peripheral |
| Effect on GH synthesis |
Yes (CREB/Pit-1) |
Primarily release |
| Appetite effect |
Minimal |
Variable (GHRP-6 > Ipamorelin) |
| Cortisol/Prolactin |
Minimal effect |
Variable by compound |
| Oral availability |
No (peptides) |
MK-677 is oral |
| Examples |
Sermorelin, CJC-1295, Tesamorelin |
Ipamorelin, GHRP-2, GHRP-6, MK-677 |
Key Takeaways
-
GHRH and GHRP are both "secretagogues" but use completely different receptors and signaling pathways - This isn't just academic; it explains their different effects and why they synergize.
-
GHRH analogs work through GHRHR → cAMP/PKA, providing relatively "physiologic" pituitary stimulation with minimal peripheral effects.
-
GHRPs and ghrelin mimetics work through GHSR → calcium/PKC, with broader tissue distribution explaining appetite and other effects.
-
Stacking GHRH + GHRP produces synergistic GH release by activating both pathways simultaneously - this is well-documented in the scientific literature.
-
MK-677 is a non-peptide oral secretagogue that functions like a GHRP (GHSR agonist), not like a GHRH analog.
-
Individual GHRPs differ in their side-effect profiles despite acting on the same receptor - Ipamorelin is generally considered more selective with fewer off-target effects.
Understanding these mechanistic differences helps explain why these peptides have different side-effect profiles, why certain combinations are more effective than using either alone, and how to make informed decisions about which approaches might be appropriate for specific research applications.
This article is for educational and research purposes only. It is not medical advice.



