Ipamorelin is the most selective growth hormone releasing peptide (GHRP) — the "cleanest GHRP" for its minimal effects on cortisol and prolactin. Clinical development was discontinued despite positive Phase II results. This is not medical advice.
Quick Reference: Standard Protocol
| Parameter |
Detail |
| Dose |
300 mcg per injection |
| Route |
Subcutaneous injection |
| Timing |
AM and/or PM, empty stomach |
| Frequency |
5 days on, 2 days off |
| Cycle |
8 weeks on, 8 weeks off |
| Vial size |
10 mg |
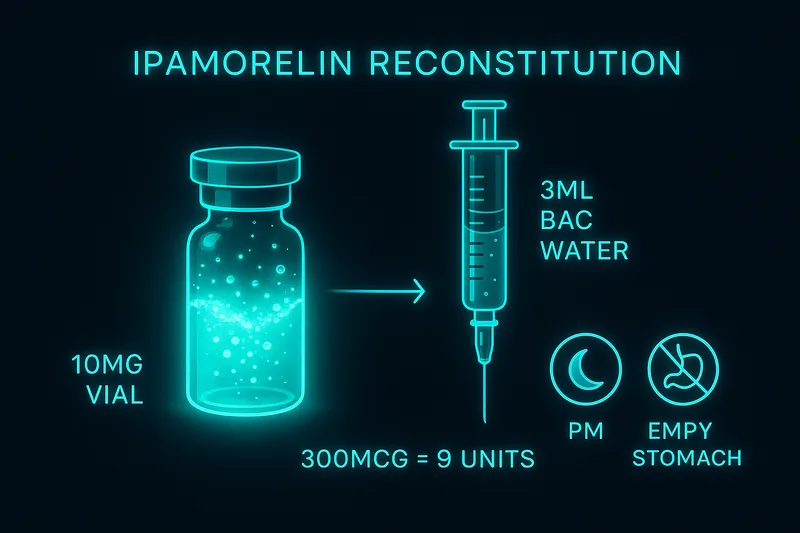
| Reconstitution |
3 mL BAC water (~3,333 mcg/mL) |
| Draw amount |
9 units on insulin syringe |
| Storage |
Refrigerate, use within 28 days |
For the full Ipamorelin peptide profile, vendor pricing, and stack protocols, see our Ipamorelin peptide page.
Cycling Details
The 5-on/2-off weekly pattern prevents receptor desensitization while maintaining consistent GH pulsatility. Start with 300 mcg once daily (morning fasted or before bed 3+ hours after last meal) during weeks 1-2 to assess tolerance, then optionally progress to twice daily for enhanced GH elevation.
The 8 weeks on / 8 weeks off cycle allows receptor sensitivity to fully recover. Never take with food — GH response is significantly blunted by carbohydrates and fats.
Routes of Administration
Subcutaneous (only practical route): Abdomen, love handles, or thighs. Volume is typically 0.09 mL (9 units on insulin syringe) for a 300 mcg dose. Use 29-31 gauge insulin syringe.
Why not oral? Like most GHRPs, ipamorelin is degraded by stomach acid with poor oral bioavailability. Subcutaneous was the route used in clinical trials.
Reconstitution Quick Reference
| Vial Size |
BAC Water |
Concentration |
300 mcg Dose |
| 10 mg |
3 mL |
~3,333 mcg/mL |
9 units |
Math: 10,000 mcg / 3 mL = 3,333 mcg/mL. 300 mcg / 3,333 = 0.09 mL = 9 units. Swirl gently, refrigerate, use within 28 days.
For step-by-step reconstitution instructions, see the Ipamorelin reconstitution guide.
Where These Numbers Come From
Community Ipamorelin protocols are directly informed by clinical trial data, making the dosing rationale more evidence-based than many peptides.
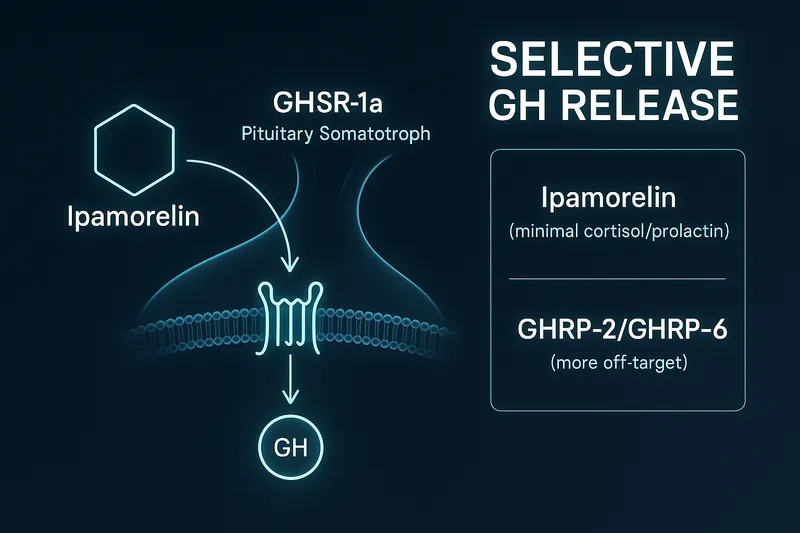
The primary clinical evidence comes from a Phase II trial for postoperative ileus using 0.01-0.1 mg/kg IV with excellent tolerability (Barlind et al., 2008). Earlier research established ipamorelin's selectivity profile — significant GH release starting at low doses with minimal cortisol/prolactin elevation compared to other GHRPs (Raun et al., 1998).
The standard 300 mcg dose comes from scaling down clinical IV doses (which were much higher) and finding the optimal GH response curve for subcutaneous use. Ipamorelin's high selectivity allows effective dosing at lower amounts than other GHRPs, and its selectivity is its primary advantage — providing clean GH pulses without hormonal side effects (Johansen et al., 1999).
Stacking Protocols
| Stack |
Ipamorelin Dose |
Partner |
Partner Dose |
Purpose |
| CJC-1295 |
300 mcg AM/PM |
CJC-1295 no DAC |
100 mcg same syringe |
Gold standard GHRH/GHRP |
| Sermorelin |
300 mcg AM/PM |
Sermorelin |
Per protocol |
All-natural GH stimulation |
| GHRP rotation |
300 mcg (weeks 1-4) |
GHRP-2 100 mcg (weeks 5-8) |
— |
Prevent desensitization |
Ipamorelin and CJC-1295 can be mixed in the same syringe. Always maintain fasted state for optimal GH response. Start separately to assess individual response before combining.
Side Effects & Safety
- Injection site redness — mild, transient
- Mild water retention — less than other GHRPs
- Increased appetite — slight, not the intense hunger of GHRP-6
- Mild headache — typically first week only
- No cortisol elevation — unlike GHRP-2/GHRP-6
- No prolactin increase — unlike hexarelin
- Active cancer — GH elevation could theoretically promote tumor growth; avoid
Frequently Asked Questions
What is the standard Ipamorelin dose?
300 mcg per injection (9 units from a 10 mg vial with 3 mL BAC water), taken 1-2 times daily on an empty stomach, 5 days on/2 days off, cycled 8 weeks on/8 weeks off.
Why is Ipamorelin considered the 'cleanest' GHRP?
Highest selectivity for growth hormone release with minimal cortisol and prolactin elevation. Clean GH pulses without the hormonal side effects seen with GHRP-2, GHRP-6, or hexarelin.
Should I take Ipamorelin once or twice daily?
Both work. Once daily (AM or PM) is simpler and cost-effective. Twice daily provides more consistent GH elevation but doubles cost. Start with once daily and assess response.
How long should an Ipamorelin cycle last?
8 weeks on, 8 weeks off, with 5 days on/2 days off each week. This prevents receptor desensitization while allowing consistent progress.
Can Ipamorelin be taken with food?
No — always on an empty stomach. Carbohydrates and fats significantly blunt the GH response. Wait 2+ hours after eating or take first thing in the morning fasted.
How do I reconstitute Ipamorelin?
Add 3 mL bacteriostatic water to a 10 mg vial (~3,333 mcg/mL). 300 mcg = 9 units on insulin syringe. Swirl gently, refrigerate, use within 28 days.
References
| Citation |
Topic |
PMID |
| Barlind et al., Clinical Drug Investigation (2008) |
Phase II postoperative ileus trial, safety profile |
18695216 |
| Raun et al., European Journal of Endocrinology (1998) |
GH release selectivity, minimal cortisol/prolactin effects |
9849822 |
| Johansen et al., Growth Hormone & IGF Research (1999) |
Selectivity profile vs other GHRPs |
10372149 |
For educational and research purposes only. This is not medical advice. Ipamorelin has clinical trial safety data but is not FDA-approved for any indication.


