 Tesofensine is one of the most potent weight loss compounds ever studied in clinical trials — producing 10–13% body weight loss in just 24 weeks during Phase 2 testing. That's roughly double what older anti-obesity drugs like sibutramine achieved, and it does it through a completely different mechanism than the GLP-1 receptor agonists dominating today's market.
Tesofensine is one of the most potent weight loss compounds ever studied in clinical trials — producing 10–13% body weight loss in just 24 weeks during Phase 2 testing. That's roughly double what older anti-obesity drugs like sibutramine achieved, and it does it through a completely different mechanism than the GLP-1 receptor agonists dominating today's market.
But here's the thing you need to know upfront: tesofensine is not a peptide. It's a small-molecule drug — a triple monoamine reuptake inhibitor that affects serotonin, norepinephrine, and dopamine. You'll find it sold by many of the same vendors that carry research peptides, which is why it shows up in peptide-related searches. This guide treats it honestly: what the clinical data actually shows, where the research stands, and what the risks are.
Table of Contents
What Is Tesofensine?
Tesofensine (NS 2330) is a synthetic small-molecule compound originally developed by NeuroSearch A/S, a Danish pharmaceutical company. It was initially investigated as a treatment for Parkinson's disease and Alzheimer's disease in the early 2000s. During those neurological trials, researchers noticed a striking and consistent side effect: significant weight loss among participants [1, 2].
This serendipitous discovery redirected tesofensine's development toward obesity treatment. The compound entered Phase 2 clinical trials specifically for weight loss, producing some of the most impressive results seen in anti-obesity drug development at that time [1].
Tesofensine is not a peptide. This distinction matters for credibility and for understanding how it works. Peptides like semaglutide and tirzepatide are large amino acid chains that bind to specific hormone receptors (GLP-1, GIP). Tesofensine is a small synthetic molecule — chemically closer to antidepressants like bupropion than to any peptide. It appears in the peptide vendor ecosystem because research compound suppliers often carry both peptides and small molecules, and because the weight loss community groups them together. But pharmacologically, they are completely different classes of compounds.
Chemically, tesofensine belongs to the phenyltropane class of compounds. Its molecular formula is C₁₇H₂₃Cl₂NO and it has a molecular weight of approximately 328.3 g/mol. It is administered orally as a once-daily tablet — no injections required, which distinguishes it from most peptide-based weight loss compounds.
Mechanism of Action
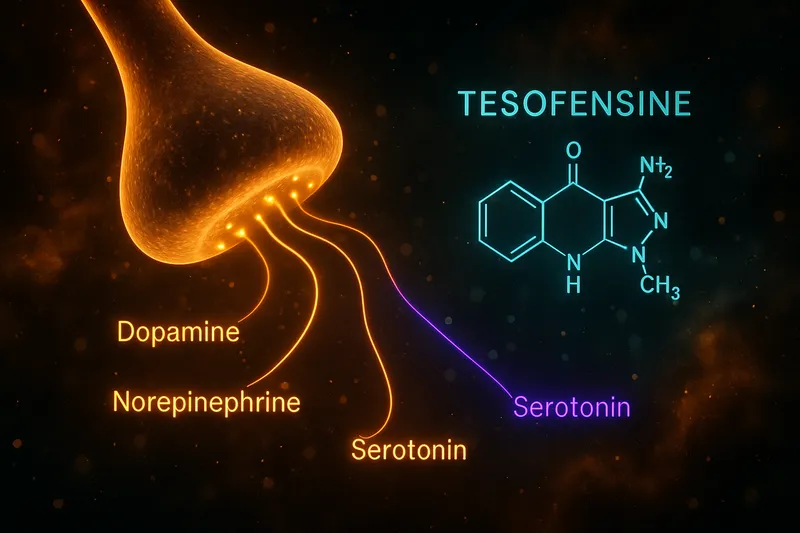
Tesofensine works as a triple monoamine reuptake inhibitor (TRI), blocking the reuptake of three key neurotransmitters simultaneously [1, 3]:
- Serotonin (5-HT) — Increases satiety signaling and reduces food cravings
- Norepinephrine (NE) — Enhances thermogenesis and resting metabolic rate
- Dopamine (DA) — Modulates reward pathways and reduces hedonic eating
This triple-action mechanism is what makes tesofensine distinctive. Most centrally acting anti-obesity drugs target only one or two of these pathways. Sibutramine (withdrawn in 2010) inhibited serotonin and norepinephrine reuptake but had minimal dopamine effects. Bupropion/naltrexone (Contrave) affects dopamine and norepinephrine but works through different receptor mechanisms. Tesofensine hits all three simultaneously.
How the Triple Mechanism Drives Weight Loss
PET imaging studies in humans have confirmed that tesofensine occupies dopamine, serotonin, and norepinephrine transporters at clinically relevant doses [4]. At the 0.5 mg dose used in Phase 2 trials, dopamine transporter occupancy was measured at approximately 30–50%, which falls in a range that suppresses appetite without producing the euphoric or stimulant-like effects associated with higher occupancy levels [4].
The combined effect produces weight loss through two complementary pathways:
Appetite Suppression: The serotonergic and dopaminergic effects reduce both homeostatic hunger (the biological drive to eat) and hedonic hunger (eating for pleasure/reward). Clinical data showed that tesofensine-treated subjects reported reduced hunger, increased fullness, and decreased desire to eat compared to placebo — effects that were sustained throughout the 24-week trial period [5, 6]. Animal studies confirmed that tesofensine reverses the low forebrain dopamine levels characteristic of diet-induced obesity, which may help normalize the dysregulated reward signaling that drives overeating [7].
Metabolic Enhancement: The noradrenergic component increases sympathetic nervous system activity, boosting resting energy expenditure. A controlled metabolic study by Sjödin et al. found that tesofensine 0.5 mg increased 24-hour energy expenditure and fat oxidation in overweight men, independent of changes in physical activity [3]. This means tesofensine doesn't just reduce intake — it also increases the calories your body burns at rest.
Animal studies using diet-induced obese rats have further characterized this dual mechanism, showing that tesofensine reduces food intake while simultaneously altering striatal dopamine D2/D3 receptor availability — a neurobiological marker associated with obesity and compulsive eating behavior [8].
How This Differs from GLP-1 Agonists
The mechanism is fundamentally different from GLP-1 receptor agonists like semaglutide. GLP-1 drugs work primarily through gut-brain signaling — they mimic incretin hormones, slow gastric emptying, and reduce appetite through vagal nerve pathways. Tesofensine works entirely within the central nervous system, directly modulating neurotransmitter levels. This means the two approaches could theoretically be complementary, though no combination studies have been published.
Clinical Trial Data
The Landmark Phase 2 Trial (TIPO-1)
The most important study on tesofensine for weight loss is the Phase 2 TIPO-1 trial published by Astrup et al. in The Lancet in 2008 [1]. This was a randomized, double-blind, placebo-controlled, multicenter trial conducted across Denmark and several European sites.
Study Design:
- Participants: 203 obese adults (BMI 30–40 kg/m²)
- Duration: 24 weeks
- Groups: Placebo, tesofensine 0.25 mg, 0.5 mg, and 1.0 mg (once daily)
- Diet: All groups followed a mildly hypocaloric diet (300 kcal/day deficit)
Results:
| Dose |
Mean Weight Loss |
Placebo-Subtracted |
| Placebo |
-2.0% |
— |
| 0.25 mg |
-4.5% |
-2.5% |
| 0.5 mg |
-9.2% |
-7.2% |
| 1.0 mg |
-10.6% |
-8.6% |
The 0.5 mg and 1.0 mg doses produced statistically significant weight loss compared to placebo (p<0.0001 for both). The weight loss was dose-dependent, with the 0.5 mg dose achieving roughly double the efficacy of any other anti-obesity drug available at that time [1].
Body Composition: Importantly, the study also measured body composition using DEXA scanning. The weight loss was predominantly fat mass, with relative preservation of lean body mass — a favorable profile compared to many weight loss interventions where muscle loss can be significant [1].
Quality of Life: Tesofensine-treated participants also showed significant improvements in quality of life measures, including physical functioning, self-esteem, and sexual life scores [1].
Important Caveat: Expression of Concern
It's essential to note that The Lancet published an expression of concern regarding the Astrup 2008 paper in 2013 [9]. This was followed by correspondence from the original authors regarding under-reporting of adverse effects [10]. The expression of concern related to potential issues with the completeness of adverse event reporting in the original trial, not to the efficacy data itself. This is a legitimate concern that anyone evaluating tesofensine should be aware of, and it has likely contributed to the compound's complicated regulatory history.
Beyond the primary efficacy trial, several supporting studies have characterized tesofensine's effects:
Sjödin et al. (2010) conducted a controlled metabolic chamber study in 32 overweight/moderately obese men. They found that tesofensine 0.5 mg significantly increased 24-hour energy expenditure (+6.1%) and fat oxidation compared to placebo after 14 days of treatment [3]. This confirmed that the weight loss mechanism involves both reduced intake and increased metabolic rate.
Gilbert et al. (2012) performed a secondary analysis of appetite data from the Phase 2 trial, confirming that tesofensine 0.5 mg and 1.0 mg significantly reduced subjective hunger ratings, increased fullness, and decreased prospective food consumption compared to placebo [5]. The appetite-suppressing effects appeared within the first two weeks and were maintained throughout the study.
PET Imaging and Transporter Occupancy
Appel et al. (2014) used PET imaging to measure tesofensine's dopamine transporter (DAT) occupancy in the human brain [4]. They found that at clinically relevant doses, tesofensine occupied approximately 30–50% of DAT — sufficient for appetite suppression but below the threshold typically associated with abuse liability or euphoric effects. This study was important for characterizing the compound's therapeutic window and addressing concerns about its stimulant-like potential.
Abuse Potential Assessment
Schoedel et al. (2010) conducted a formal abuse liability study comparing tesofensine to d-amphetamine in recreational stimulant users [11]. While tesofensine did produce some subjective effects (e.g., increased alertness), these were significantly less than d-amphetamine at comparable doses. The authors concluded that tesofensine's abuse potential appeared lower than classic psychostimulants, consistent with its moderate transporter occupancy profile.
Preclinical Evidence
Multiple animal studies have corroborated the clinical findings:
-
Hansen et al. (2013) showed that tesofensine produced dose-dependent weight loss in diet-induced obese rats, reversed low forebrain dopamine levels, and suppressed food intake without affecting locomotor activity — suggesting the weight loss isn't simply due to stimulant-driven hyperactivity [7].
-
Van de Giessen et al. (2012) demonstrated that tesofensine reduced food intake and body weight in obese rats while also decreasing striatal D2/D3 receptor availability, suggesting it may normalize the dopaminergic dysfunction associated with obesity [8].
Dosing Protocols
What Clinical Trials Used
The Phase 2 trial tested three doses [1]:
-
0.25 mg once daily — Produced modest weight loss (~4.5%). Below the threshold for clinically meaningful efficacy in most subjects. This dose did have a favorable side effect profile.
-
0.5 mg once daily — The "sweet spot" dose. Produced ~9.2% body weight loss with a manageable side effect profile. This is the dose that generated the most interest for further development and is most commonly referenced in the literature [1, 2].
-
1.0 mg once daily — Produced the most weight loss (~10.6%) but with a notably higher incidence of side effects, particularly elevated heart rate and mood disturbances. The incremental weight loss over the 0.5 mg dose was modest relative to the increase in adverse effects [1].
Timing and Administration
In the clinical trials, tesofensine was administered as a single oral dose taken once daily, typically in the morning. The compound has a long half-life (approximately 220 hours, or roughly 8–9 days), which means steady-state blood levels are reached after approximately 5–6 weeks of daily dosing [4, 11].
This pharmacokinetic profile has important implications:
- Slow onset: Full effects may not be apparent for several weeks
- Slow offset: Effects (and side effects) persist for days after discontinuation
- Accumulation: The long half-life means the drug accumulates significantly with daily dosing, so steady-state concentrations are much higher than single-dose levels
Important Dosing Considerations
There is no FDA-approved dosing protocol for tesofensine because the drug has never received regulatory approval. The dosing information above comes exclusively from clinical trial data. Research compound vendors may sell tesofensine in various formats and concentrations that differ from what was studied clinically.
The long half-life also means that dose adjustments take weeks to fully manifest. If starting at a lower dose and titrating up, a minimum of 4–6 weeks at each dose level would be needed before the full effect can be assessed — consistent with how the clinical trials managed dosing.
Anyone considering tesofensine should understand that this is an unapproved investigational compound, and self-dosing based on clinical trial protocols carries inherent risks. Physician supervision is strongly recommended, as cardiovascular monitoring (heart rate, blood pressure) was an integral part of all clinical trials.
Tesofensine vs Semaglutide and Tirzepatide
The weight loss landscape has changed dramatically since tesofensine's Phase 2 data was published in 2008. The GLP-1 receptor agonists — particularly semaglutide and tirzepatide — have set a new standard for pharmacological weight loss. Here's how tesofensine compares:
Efficacy Comparison
| Compound |
Trial |
Duration |
Mean Weight Loss |
| Tesofensine 0.5 mg |
TIPO-1 Phase 2 [1] |
24 weeks |
~9.2% |
| Tesofensine 1.0 mg |
TIPO-1 Phase 2 [1] |
24 weeks |
~10.6% |
| Semaglutide 2.4 mg |
STEP 1 Phase 3 [12] |
68 weeks |
~14.9% |
| Tirzepatide 15 mg |
SURMOUNT-1 Phase 3 [13] |
72 weeks |
~22.5% |
Critical context: These numbers aren't directly comparable. The tesofensine data comes from a smaller Phase 2 trial (203 participants) over just 24 weeks. The semaglutide and tirzepatide data come from much larger Phase 3 trials (1,961 and 2,539 participants) over 68–72 weeks. Weight loss with GLP-1 drugs typically continues beyond 24 weeks, so the GLP-1 advantage may be larger than the raw numbers suggest. Conversely, tesofensine's weight loss trajectory at 24 weeks was still trending downward, so a longer trial might have shown greater total loss.
Mechanism Differences
| Feature |
Tesofensine |
GLP-1 Agonists |
| Drug Class |
Triple monoamine reuptake inhibitor |
Incretin mimetics |
| Primary Target |
CNS neurotransmitters (5-HT, NE, DA) |
GLP-1 receptors (gut, brain, pancreas) |
| Route |
Oral (once daily) |
Subcutaneous injection (weekly) |
| Appetite Mechanism |
Central neurotransmitter modulation |
Gut-brain axis, gastric slowing |
| Metabolic Effect |
Increased resting energy expenditure |
Improved insulin sensitivity, glucose control |
| GI Side Effects |
Minimal |
Nausea, vomiting common (20-40%) |
| CV Concerns |
Elevated heart rate |
CV protective (proven in trials) |
| Regulatory Status |
Not approved anywhere |
FDA approved |
Potential for Combination
Because tesofensine and GLP-1 agonists work through entirely different mechanisms, there's theoretical interest in combination approaches. Tesofensine addresses the central dopaminergic reward pathways that GLP-1 drugs don't directly target, while GLP-1 drugs address peripheral metabolic signaling and gastric motility that tesofensine doesn't affect.
However, no clinical combination studies exist. This remains purely theoretical, and combining an unapproved investigational compound with FDA-approved medications would carry unknown risk profiles. The cardiovascular effects of tesofensine (increased heart rate) could potentially counteract the cardiovascular benefits of GLP-1 agonists.
For those interested in approved multi-target approaches to weight loss, compounds like retatrutide — which targets GLP-1, GIP, and glucagon receptors simultaneously — represent a different kind of "stacking" approach within the peptide class. You can also explore our peptide stacking guide for more on combining research compounds.
Practical Advantages of Tesofensine
Despite weaker efficacy data compared to modern GLP-1 drugs, tesofensine does have some theoretical advantages:
- Oral dosing — No injections required, which improves convenience and compliance
- Different side effect profile — Minimal GI side effects (no nausea, vomiting, or diarrhea), which are the most common reasons patients discontinue GLP-1 therapy
- Dopaminergic component — May specifically address reward-driven overeating and food addiction in ways that GLP-1 drugs don't
- Cost — As an unapproved research compound, tesofensine is typically far less expensive than branded GLP-1 medications (though with all the caveats about quality, purity, and safety that come with unregulated compounds)
Side Effects and Safety Profile
The safety data for tesofensine comes primarily from the Phase 2 TIPO-1 trial and supporting studies. Several review papers have also analyzed its safety profile in the context of centrally acting anti-obesity drugs [14, 15, 16].
Common Side Effects (from Phase 2 trial data)
Dose-dependent effects at 0.5 mg (most studied dose):
-
Dry mouth — Reported by approximately 30-35% of participants. The most common side effect, likely related to noradrenergic and serotonergic activity. Generally mild and well-tolerated.
-
Insomnia — Reported by approximately 15-20% of participants. Consistent with the noradrenergic and dopaminergic stimulation. Morning dosing was used in trials to minimize this effect.
-
Constipation — Reported by approximately 10-15% of participants. Related to reduced GI motility from sympathetic nervous system activation.
-
Hard stools — Related to constipation, reported at similar rates.
-
Elevated heart rate — Mean increase of 7.4 bpm at 0.5 mg and 8.1 bpm at 1.0 mg compared to placebo [1]. This was the most clinically concerning side effect and a major factor in the compound's regulatory challenges.
Cardiovascular Concerns
The heart rate increase was the primary safety signal that complicated tesofensine's development [1, 14]. While the mean increase was moderate (7-8 bpm), individual responses varied considerably. Some participants experienced clinically significant tachycardia.
This cardiovascular profile contrasts unfavorably with GLP-1 agonists, which have demonstrated cardiovascular protective effects in large outcomes trials. The history of centrally acting anti-obesity drugs and cardiovascular risk is cautionary — sibutramine (a dual serotonin-norepinephrine reuptake inhibitor) was withdrawn from the market in 2010 after the SCOUT trial showed increased cardiovascular events [15].
Tesofensine has a mechanistic similarity to sibutramine (both inhibit monoamine reuptake), though tesofensine additionally targets dopamine. The cardiovascular safety profile would need to be thoroughly evaluated in large Phase 3 trials — which have not been completed.
Blood Pressure Effects
In the Phase 2 trial, blood pressure changes were modest. Mean systolic blood pressure showed small, non-significant increases, while diastolic blood pressure was largely unchanged [1]. However, individual responses varied, and the trial was not powered or designed to fully characterize blood pressure effects in high-risk populations.
Mood and Psychiatric Effects
Given that tesofensine modulates serotonin, norepinephrine, and dopamine — the same neurotransmitters targeted by antidepressants and stimulants — psychiatric effects are a legitimate concern. Nathan et al. (2011) reviewed the neuropsychiatric adverse effects of centrally acting anti-obesity drugs, including tesofensine, noting that mood elevation, anxiety, and depressive symptoms have been observed with this drug class [14].
In the Phase 2 trial, mood-related adverse events were more common in the 1.0 mg group than the 0.5 mg group, which was one factor supporting the 0.5 mg dose as the optimal balance of efficacy and tolerability [1].
The Expression of Concern
As noted in the clinical trials section, The Lancet published an expression of concern in 2013 regarding the original Astrup et al. trial [9], specifically related to the completeness of adverse event reporting. The original authors responded with a letter addressing the under-reporting concerns [10]. This unresolved issue adds uncertainty to the safety database and is another reason the compound has not progressed to approval.
Current Regulatory Status
Development History
Tesofensine's regulatory journey has been turbulent:
- Early 2000s: Developed by NeuroSearch A/S (Denmark) for Parkinson's disease
- 2006-2007: Pivoted to obesity indication after weight loss observed in neurological trials
- 2008: Phase 2 results published in The Lancet [1], generating significant pharmaceutical interest
- 2010-2011: NeuroSearch attempted to license or partner for Phase 3 development
- 2013: Lancet expression of concern published [9]; NeuroSearch entered financial difficulties
- 2016: Rights transferred to Saniona A/S (Denmark), which licensed them to Medican (Mexico) for Latin American development
- 2019-present: Medican (now rebranded) has pursued limited development in Mexico; no Phase 3 results published in peer-reviewed journals
Current Status (as of early 2026)
Tesofensine is NOT approved by the FDA, EMA, or any major regulatory agency for any indication. It has not completed Phase 3 clinical trials. The compound is available only as:
- A research chemical sold by peptide/research compound vendors
- A compounded preparation available through some telehealth and compounding pharmacy channels (in a legal gray area)
The compound has no official prescribing information, no standardized manufacturing process for consumer products, and no post-marketing safety surveillance. Anyone using tesofensine is doing so entirely outside of approved medical practice.
Why Development Stalled
Several factors contributed to tesofensine's failure to reach market:
- Cardiovascular safety concerns — The heart rate increase made regulators cautious, especially after sibutramine's withdrawal
- The Lancet expression of concern — Undermined confidence in the primary efficacy/safety dataset
- NeuroSearch's financial collapse — The small Danish company couldn't fund Phase 3 trials independently
- Competition from GLP-1 drugs — The approval of liraglutide (2014) and semaglutide (2021) for obesity reduced commercial interest in tesofensine
- Regulatory climate — Post-fen-phen, post-sibutramine, regulators demand extensive cardiovascular outcome data for anti-obesity drugs, which is extremely expensive to generate
Frequently Asked Questions
Is tesofensine a peptide?
No. Tesofensine is a small-molecule synthetic drug (a phenyltropane derivative), not a peptide. It has no amino acid sequence and does not bind to peptide hormone receptors. It appears in peptide vendor catalogs because the same companies that sell research peptides also carry small-molecule research compounds. For an introduction to what peptides actually are, see our guide to peptides.
How does tesofensine compare to semaglutide for weight loss?
Semaglutide has stronger clinical evidence — large Phase 3 trials showing ~15% weight loss over 68 weeks with FDA approval and demonstrated cardiovascular safety. Tesofensine's Phase 2 data showed ~9-11% loss over 24 weeks but has never completed Phase 3 trials. They work through completely different mechanisms (central monoamine reuptake inhibition vs. GLP-1 receptor agonism). See our semaglutide vs tirzepatide comparison for more on approved options.
What dose of tesofensine was used in clinical trials?
The Phase 2 trial tested 0.25 mg, 0.5 mg, and 1.0 mg once daily [1]. The 0.5 mg dose is generally considered the best balance of efficacy and tolerability, producing ~9.2% body weight loss over 24 weeks with fewer side effects than the 1.0 mg dose.
Is tesofensine FDA approved?
No. Tesofensine has never been approved by the FDA, EMA, or any major regulatory authority. It has not completed Phase 3 clinical trials. It is only available as a research compound or through compounding pharmacies.
Can you combine tesofensine with GLP-1 drugs?
There are no published clinical studies on combining tesofensine with semaglutide, tirzepatide, or any other GLP-1 agonist. While the different mechanisms suggest theoretical complementarity, the safety of such combinations is unknown. The cardiovascular effects of tesofensine (increased heart rate) are a particular concern in any combination context.
What are the main side effects of tesofensine?
The most commonly reported side effects in clinical trials were dry mouth (~30-35%), insomnia (~15-20%), constipation (~10-15%), and elevated heart rate (mean increase of ~7-8 bpm) [1]. The heart rate increase is the most clinically significant concern. Side effects were dose-dependent, with the 1.0 mg dose showing more adverse events than the 0.5 mg dose.
How long does it take for tesofensine to work?
Due to its long half-life (~220 hours), tesofensine takes approximately 5–6 weeks to reach steady-state blood levels [4]. Initial appetite-suppressing effects may be noticed within 1–2 weeks, but maximum efficacy requires several weeks of consistent daily dosing. In the Phase 2 trial, weight loss continued throughout the full 24-week treatment period [1].
Why isn't tesofensine available as a prescription drug?
Primarily because NeuroSearch, the company that developed it, ran out of funding before completing Phase 3 trials. The cardiovascular safety profile also made regulatory approval challenging in the post-sibutramine environment. Current rights holders have pursued limited development in Latin America, but no major regulatory submission has been made.
Is tesofensine safe?
The Phase 2 safety data is limited (203 participants, 24 weeks) and the expression of concern regarding adverse event reporting adds uncertainty [9, 10]. The cardiovascular effects (elevated heart rate) are a legitimate concern, especially for individuals with pre-existing cardiac conditions. Without Phase 3 data and post-marketing surveillance, the full safety profile of tesofensine remains incompletely characterized. Physician supervision is strongly recommended for anyone considering this compound.
Can tesofensine help with food addiction or emotional eating?
The dopaminergic component of tesofensine's mechanism is theoretically relevant to reward-driven eating behaviors. Animal studies have shown that tesofensine reverses low forebrain dopamine levels in diet-induced obesity and reduces striatal D2/D3 receptor availability [7, 8]. Clinical appetite data showed reduced desire to eat and reduced hunger [5]. However, tesofensine has not been specifically studied for food addiction or binge eating disorder in clinical trials.
References
-
Astrup A, Madsbad S, Breum L, Jensen TJ, Kroustrup JP, Larsen TM. Effect of tesofensine on bodyweight loss, body composition, and quality of life in obese patients: a randomised, double-blind, placebo-controlled trial. Lancet. 2008;372(9653):1906-1913. PMID: 18950853
-
Doggrell SA. Tesofensine — a novel potent weight loss medicine. Expert Opin Investig Drugs. 2009;18(7):1043-1046. PMID: 19548858
-
Sjödin A, Gasteyger C, Nielsen AL, et al. The effect of the triple monoamine reuptake inhibitor tesofensine on energy metabolism and appetite in overweight and moderately obese men. Int J Obes (Lond). 2010;34(11):1634-1643. PMID: 20479765
-
Appel L, Bergström M, Buus Lassen J, Långström B. Tesofensine, a novel triple monoamine re-uptake inhibitor with anti-obesity effects: dopamine transporter occupancy as measured by PET. Eur Neuropsychopharmacol. 2014;24(2):251-261. PMID: 24239329
-
Gilbert JA, Gasteyger C, Raben A, Meier DH, Astrup A, Sjödin A. The effect of tesofensine on appetite sensations. Obesity (Silver Spring). 2012;20(3):553-561. PMID: 21720440
This article is for educational and informational purposes only. Tesofensine is not approved by the FDA or any major regulatory agency. It is not intended to diagnose, treat, cure, or prevent any disease. Always consult a qualified healthcare provider before using any research compound. The Peptide Catalog does not sell tesofensine or any other research compounds.
 Tesofensine is one of the most potent weight loss compounds ever studied in clinical trials — producing 10–13% body weight loss in just 24 weeks during Phase 2 testing. That's roughly double what older anti-obesity drugs like sibutramine achieved, and it does it through a completely different mechanism than the GLP-1 receptor agonists dominating today's market.
Tesofensine is one of the most potent weight loss compounds ever studied in clinical trials — producing 10–13% body weight loss in just 24 weeks during Phase 2 testing. That's roughly double what older anti-obesity drugs like sibutramine achieved, and it does it through a completely different mechanism than the GLP-1 receptor agonists dominating today's market.
