 Thymosin Beta-4 (TB-4) is a 43-amino-acid peptide found in virtually every human cell except red blood cells. It's the body's most abundant actin-sequestering protein — a fundamental building block for cell movement, tissue repair, and wound healing. Since the late 1990s, a growing body of preclinical research has demonstrated TB-4's ability to accelerate recovery across a remarkable range of injury types.
Thymosin Beta-4 (TB-4) is a 43-amino-acid peptide found in virtually every human cell except red blood cells. It's the body's most abundant actin-sequestering protein — a fundamental building block for cell movement, tissue repair, and wound healing. Since the late 1990s, a growing body of preclinical research has demonstrated TB-4's ability to accelerate recovery across a remarkable range of injury types.
This guide examines what the published research actually shows about TB-4 for injury recovery. We cover every major tissue type studied — from skin wounds and corneal damage to cardiac infarction and spinal cord injury. Every claim is linked to published data. No anecdotes, no hype — just the science.
Table of Contents
Before examining specific injury types, it's important to understand the mechanisms that make TB-4 relevant to recovery across so many different tissues. TB-4 doesn't treat a single condition — it enhances the fundamental repair processes that all injured tissues share.
1. Cell Migration
TB-4's primary intracellular function is sequestering G-actin monomers, regulating the polymerization of actin filaments that drive cell movement. When tissue is damaged, repair cells (fibroblasts, endothelial cells, keratinocytes, stem cells) must migrate to the injury site. TB-4 accelerates this migration by modulating the actin cytoskeleton — the cellular machinery that powers cell motility (Hannappel, 2007).
2. Anti-Inflammation
TB-4 suppresses NF-κB — the master transcription factor controlling inflammatory gene expression. By reducing NF-κB activation, phosphorylation, and nuclear translocation, TB-4 dampens the production of pro-inflammatory cytokines (TNF-α, IL-1β, IL-6) that can prolong tissue damage beyond the initial injury (Sosne et al., 2007).
3. Angiogenesis
New blood vessel formation is essential for delivering oxygen, nutrients, and repair cells to damaged tissue. TB-4 promotes angiogenesis through its actin-binding domain, which stimulates endothelial cell proliferation, migration, and tube formation. This is particularly critical in tissues with poor baseline vascularity, like tendons and cartilage (Philp et al., 2003).
4. Stem and Progenitor Cell Activation
TB-4 activates resident stem and progenitor cell populations in multiple tissues. In skin, it mobilizes hair follicle bulge stem cells. In the heart, it reactivates epicardium-derived progenitor cells. In the brain, it promotes neurogenesis from endogenous neural progenitors. This stem cell activation distinguishes TB-4 from most other healing peptides (Philp et al., 2004).
Dermal and Skin Wound Recovery
Skin wound healing was the first injury type where TB-4's therapeutic potential was demonstrated, and it remains one of the best-studied applications.
Full-Thickness Skin Wounds
The foundational study by Malinda et al. (1999) established TB-4 as a wound healing agent. In a rat full-thickness wound model, topical TB-4 application significantly accelerated wound closure compared to saline controls. Treated wounds showed:
- Faster wound contraction — measurable within the first week
- Increased angiogenesis — new blood vessel formation at wound margins
- Enhanced collagen deposition — more organized extracellular matrix
- Accelerated re-epithelialization — faster coverage of the wound bed
The effect was dose-dependent and consistent across multiple wound sizes (Malinda et al., 1999).
Diabetic Wound Healing
Chronic non-healing wounds are a major complication of diabetes. Standard wound care often fails because diabetic tissue has impaired angiogenesis, chronic inflammation, and reduced cell migration — exactly the processes TB-4 enhances.
Kim et al. (2014) studied TB-4 in db/db diabetic mice with dermal burn wounds. TB-4 treatment significantly improved wound closure in these metabolically compromised animals. The mechanism involved downregulation of RAGE (receptor for advanced glycation end products), a key mediator of diabetic wound pathology. TB-4 also increased angiogenesis and reduced inflammatory infiltration at the wound site (Kim et al., 2014).
This study is particularly relevant because diabetic wounds represent a clinical scenario where standard healing is impaired — and TB-4 addressed the specific pathological mechanisms responsible.
Dermal Healing: Comprehensive Review
Kleinman and Sosne (2016) published a comprehensive review of TB-4 in dermal healing, summarizing evidence from multiple animal models including aged mice, diabetic mice, and standard wound models. They noted that TB-4 is a naturally occurring regenerative protein found in body fluids and inside cells, and that no other single agent has been identified that significantly accelerates chronic dermal wound repair in humans. The review concluded that TB-4 addresses multiple overlapping deficits in wound healing simultaneously — migration, inflammation, angiogenesis, and extracellular matrix deposition (Kleinman & Sosne, 2016).
Enhanced Wound Healing with Dimeric TB-4
Xu et al. (2013) developed a novel dimeric form of TB-4 with enhanced biological activities. In dermal wound models, this dimeric TB-4 accelerated wound closure even more than the standard monomeric form, suggesting that TB-4's wound healing effects are dose- and structure-dependent. The dimeric form showed improved cell migration stimulation and accelerated the rate of wound closure compared to monomeric TB-4 (Xu et al., 2013).
Corneal and Ocular Injury
Corneal healing is one of TB-4's most clinically advanced applications, with Phase 2/3 clinical trials conducted using topical TB-4 formulations.
Alkali Burn Injury
Sosne et al. (2002) studied TB-4 in a severe corneal injury model — alkali burns, which cause extensive epithelial damage, stromal inflammation, and can lead to permanent vision loss. Mice treated with topical TB-4 twice daily showed:
- Accelerated epithelial healing — faster coverage of the denuded corneal surface
- Reduced inflammation — fewer infiltrating polymorphonuclear leukocytes
- Improved corneal clarity — less haze and scarring
The effect was significant within 7 days and continued to improve through the observation period (Sosne et al., 2002).
Anti-Inflammatory Mechanism in the Cornea
The mechanism behind TB-4's corneal healing involves direct suppression of NF-κB in corneal epithelial cells. Sosne et al. (2007) demonstrated that TB-4 treatment reduced NF-κB protein levels, inhibited TNF-α-induced NF-κB phosphorylation, and blocked nuclear translocation of the p65 subunit — effectively shutting down the inflammatory cascade that damages corneal tissue after injury (Sosne et al., 2007).
This dual action — promoting healing while simultaneously reducing the inflammation that impairs healing — makes TB-4 particularly effective for corneal injuries.
From Bench to Bedside
Sosne (2018) reviewed the journey of TB-4 from basic corneal research to Phase 3 clinical trials. The topical TB-4 formulation (RGN-259) has been evaluated in clinical trials for dry eye disease and neurotrophic keratopathy — conditions where the corneal surface fails to heal properly. Earlier phase trials showed promising results in improving corneal staining scores (a measure of surface damage) and reducing symptoms (Sosne, 2018).
The ocular application of TB-4 represents the closest translation from animal research to human clinical use and provides the best evidence for TB-4's safety and efficacy in humans.
Comprehensive Ocular Review
Sosne et al. (2016) provided an extensive review of TB-4 for neurotrophic keratopathy, dry eye, and ocular surface diseases. They noted that existing treatments often fail to address the underlying disease process or promote mechanisms that facilitate long-term wound repair. TB-4's unique combination of anti-inflammatory, pro-migratory, and wound healing properties positions it as a potential treatment that addresses the root cause rather than just symptoms (Sosne et al., 2016).
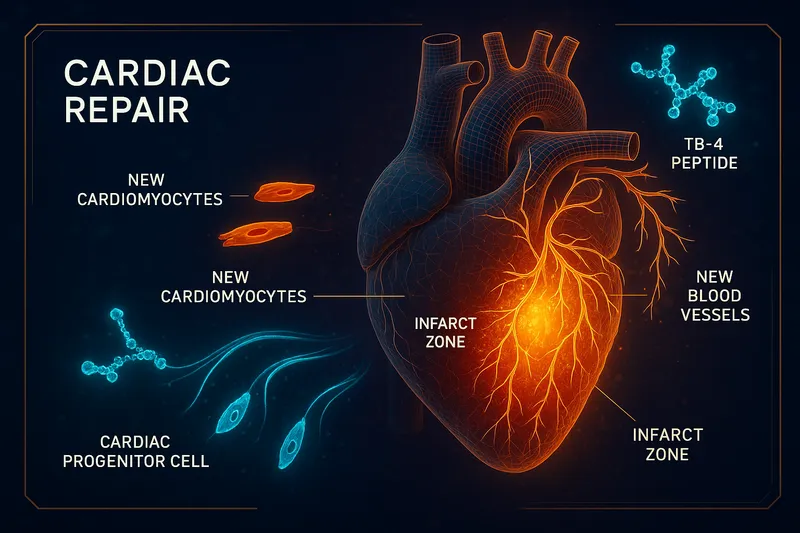
Cardiac Injury and Myocardial Infarction
Cardiac repair is arguably TB-4's most scientifically significant application. The heart has extremely limited regenerative capacity — after a heart attack, dead cardiomyocytes are replaced by non-contractile scar tissue. TB-4 research has challenged this paradigm.
The Landmark Nature Study
Bock-Marquette et al. (2004) published a groundbreaking study in Nature demonstrating that TB-4 activates integrin-linked kinase (ILK), which in turn activates the Akt survival pathway in cardiac cells. In mice with coronary artery ligation (simulating a heart attack):
- TB-4 treatment enhanced early cardiomyocyte survival — fewer heart muscle cells died in the infarct border zone
- TB-4 promoted cardiac cell migration — repair cells moved into the damaged area faster
- TB-4 improved cardiac function — echocardiography showed better heart performance
This study established the ILK-Akt pathway as TB-4's primary cardioprotective mechanism and opened the door to cardiac repair research (Bock-Marquette et al., 2004).
Cardioprotection After Myocardial Infarction
Bock-Marquette et al. (2007) expanded on the initial findings, demonstrating that TB-4 administered after myocardial infarction provided sustained cardioprotection. The peptide upregulated ILK and Akt activity in the heart, enhanced early myocyte survival, and improved cardiac function at 4 weeks post-infarction. Treated hearts showed significantly less scar tissue and better preservation of contractile function (Bock-Marquette et al., 2007).
Epicardial Progenitor Activation
Smart et al. (2007) made another critical discovery: TB-4 is essential for coronary vessel development and can reactivate the adult epicardium — the outer layer of the heart that contains dormant progenitor cells. When secreted from the myocardium, TB-4 provides a paracrine signal that:
- Promotes epicardium-derived cell (EPDC) migration inward into the heart wall
- Stimulates EPDC differentiation into vascular smooth muscle cells and endothelial cells
- Drives neovascularization — new coronary vessel formation
This mechanism is unique to TB-4 and represents a potential pathway for genuine cardiac regeneration rather than just damage limitation (Smart et al., 2007).
Cardiac Repair with Stem Cell Enhancement
Tan et al. (2021) demonstrated in a porcine (pig) model of acute myocardial infarction that TB-4 enhanced the therapeutic potential of human induced-pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs). TB-4 increased cardiac cell proliferation, improved cell engraftment at the injury site, and enhanced the reparative potency of the transplanted cells. This large animal model provides stronger translational evidence than rodent studies alone (Tan et al., 2021).
Protein Therapy for Cardiac Repair
Dubé et al. (2012) reviewed the evidence for TB-4 as a protein therapy for cardiac repair, noting the paradigm shift from cell therapy to factor-based therapy in regenerative medicine. They described TB-4's ability to inhibit myocardial cell death, stimulate vessel growth, and activate endogenous cardiac progenitors — effectively reminding the adult heart of its embryonic developmental program (Dubé et al., 2012).
Neurological Injury: Brain and Spinal Cord
The central nervous system has notoriously limited regenerative capacity. TB-4 research in neurological injury models has shown promising results across multiple injury types.
Traumatic Brain Injury (TBI)
Xiong et al. (2012) studied TB-4 in a controlled cortical impact model of TBI in rats. TB-4 treatment initiated after injury promoted:
- Neurogenesis — new neuron formation in the hippocampus and injury border zone
- Angiogenesis — new blood vessel formation in the damaged region
- Oligodendrogenesis — new myelin-forming cells to repair white matter
- Improved spatial learning — better performance on the Morris water maze
- Better sensorimotor function — improved neurological severity scores
The effects were observed through extended follow-up, suggesting TB-4 enhances endogenous neurorestorative processes rather than just providing acute neuroprotection (Xiong et al., 2012).
Spinal Cord Injury
Cheng et al. (2014) investigated TB-4 in rat spinal cord compression injury. TB-4 or saline was administered by intraperitoneal injection at multiple timepoints after injury. Results showed:
- Improved locomotor recovery — significantly better BBB (Basso-Beattie-Bresnahan) scale scores
- Better footprint analysis — improved gait coordination
- Neuroprotection — reduced neuronal loss and preserved spinal cord architecture
- Anti-inflammation — decreased inflammatory cell infiltration
- Vasculoprotection — preserved blood-spinal cord barrier integrity
Behavioral improvements were measurable by day 14 and continued through the observation period. The authors concluded that TB-4's combination of neuroprotective, anti-inflammatory, and vasculoprotective properties make it a strong candidate for SCI therapy (Cheng et al., 2014).
Stroke
Morris et al. (2010) reviewed TB-4 as a candidate for stroke treatment. After ischemic stroke, the brain mounts an endogenous repair response including neurogenesis, angiogenesis, axonal sprouting, and synaptogenesis. However, this natural response is insufficient for full recovery. TB-4's ability to enhance all of these processes makes it a logical therapeutic candidate for post-stroke neurorestoration (Morris et al., 2010).
Neuroinflammation and Neurodegeneration
Pardon (2018) reviewed TB-4's anti-inflammatory potential in the central nervous system, focusing on its implications for progressive neurodegenerative diseases. TB-4's ability to modulate microglial activation — the brain's primary inflammatory response — suggests potential applications beyond acute injury into chronic neurodegenerative conditions like Alzheimer's and Parkinson's disease. The review noted that TB-4 suppresses pro-inflammatory microglial phenotypes while potentially promoting neuroprotective microglial functions (Pardon, 2018).
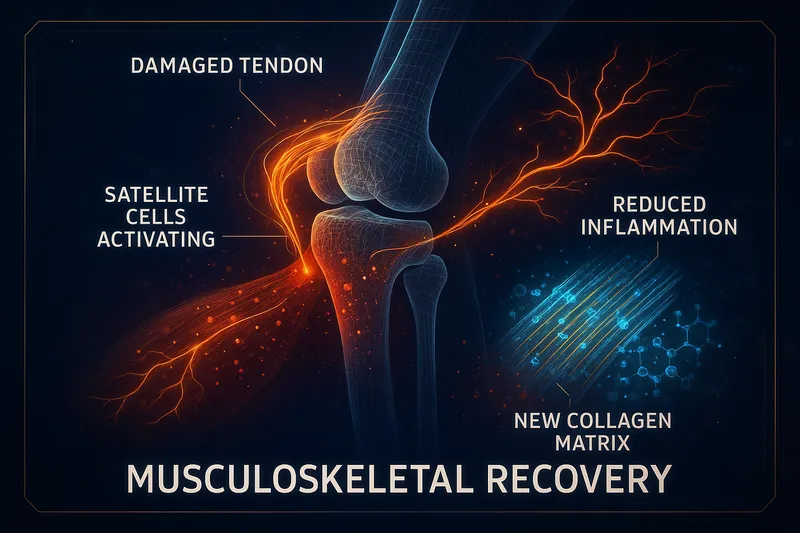
Musculoskeletal Injury
While TB-4's musculoskeletal research is less extensive than its dermal, ocular, and cardiac research, the available data shows consistent benefits in tissue types relevant to sports and orthopedic injuries.
Mechanism in Musculoskeletal Tissue
TB-4 promotes musculoskeletal recovery through the same core mechanisms active in other tissues:
- Cell migration — fibroblasts, tenocytes, and myoblasts reach the injury faster
- Angiogenesis — new blood vessel formation in poorly vascularized tissue (tendons, ligaments)
- Anti-inflammation — reduced NF-κB-mediated inflammatory damage
- Extracellular matrix remodeling — improved collagen organization over disordered scar
Philp and Kleinman (2010) reviewed multiple animal models demonstrating TB-4's tissue repair and regeneration properties, including dermal, corneal, and cardiac applications, providing the scientific foundation for clinical trials across tissue types (Philp & Kleinman, 2010).
Relevance to Tendon and Ligament Injuries
Tendons and ligaments heal slowly due to poor blood supply and low cellular density. TB-4's ability to promote angiogenesis in avascular tissue and accelerate cell migration to injury sites directly addresses the two main reasons tendon healing is slow.
While direct tendon-specific TB-4 studies are limited compared to BPC-157's extensive tendon research, TB-4's proven angiogenic and cell migration effects in multiple tissue models provide a strong mechanistic rationale for tendon healing applications.
For tendon-specific research with extensive direct evidence, see our BPC-157 Benefits Guide, which covers Achilles, MCL, and rotator cuff tendon repair data.
Hair Follicle and Skin Appendage Recovery
TB-4 promotes recovery of skin appendages — particularly hair follicles — after injury. Philp et al. demonstrated that TB-4 activates hair follicle stem cells, promoting their migration from the bulge region to the base of the follicle and stimulating differentiation into new hair-producing cells. This effect was observed in wound-adjacent skin, suggesting TB-4 may help restore normal skin architecture (including hair growth) after dermal injuries (Philp et al., 2004; Philp et al., 2007).
TB-4's Core Recovery Mechanisms
Understanding how TB-4 works at the molecular level helps explain why it's effective across such diverse injury types.
ILK-Akt Survival Pathway
TB-4 activates integrin-linked kinase (ILK), which phosphorylates Akt — a central pro-survival kinase. This pathway:
- Inhibits apoptosis — reduces programmed cell death in injured tissue
- Promotes cell survival — keeps viable cells alive in the injury border zone
- Activates downstream repair cascades — triggers pathways for proliferation and migration
This was first demonstrated in cardiac tissue but is likely active in all cell types expressing ILK (Bock-Marquette et al., 2004).
NF-κB Suppression
NF-κB is the master switch for inflammatory gene expression. TB-4 suppresses NF-κB at multiple levels — reducing protein levels, blocking phosphorylation, and preventing nuclear translocation. This provides broad-spectrum anti-inflammatory protection without complete immunosuppression (Sosne et al., 2007).
TB-4's primary intracellular role — sequestering G-actin monomers — directly controls how fast cells can reorganize their cytoskeleton and move. The actin-binding site on TB-4 has been specifically linked to its angiogenic properties, confirming that the same domain responsible for cell migration also drives new blood vessel formation (Philp et al., 2003).
Multi-System Animal Studies
Philp and Kleinman (2010) reviewed animal studies across multiple disease and repair models, concluding that TB-4's biological activities — including down-regulation of inflammatory chemokines and cytokines, promotion of cell migration, blood vessel formation, cell survival, and stem cell maturation — collectively explain its effectiveness across diverse injury types. All of these activities contribute to the multiple wound healing properties observed in animal models and support the ongoing clinical trials (Philp & Kleinman, 2010).
TB-4 vs BPC-157 for Injury Recovery
TB-4 and BPC-157 are the two most-studied healing peptides in preclinical research. They work through different mechanisms and have different strengths:
| Factor |
TB-4 |
BPC-157 |
| Size |
43 amino acids |
15 amino acids |
| Origin |
Ubiquitous intracellular protein |
Gastric juice protein fragment |
| Primary mechanism |
Actin sequestration, ILK-Akt pathway |
NO system modulation, VEGF upregulation |
| Best evidence |
Cardiac, corneal, dermal, neurological |
GI, tendon, ligament, muscle |
| Unique strength |
Stem/progenitor cell activation |
GI stability, oral bioavailability |
| Clinical trials |
Phase 2/3 (ocular) |
None (preclinical only) |
| Anti-inflammatory |
NF-κB suppression |
NO system normalization |
| Angiogenesis |
Via actin-binding domain |
Via VEGF upregulation |
When TB-4 May Be More Relevant
- Cardiac injury or repair
- Neurological injury (TBI, spinal cord, stroke)
- Conditions requiring stem cell activation
- Corneal and ocular surface damage
When BPC-157 May Be More Relevant
- Tendon and ligament injuries (most direct evidence)
- GI tract healing (ulcers, IBD, leaky gut)
- NSAID-related damage
- Muscle injuries with direct evidence
Complementary Use
Many researchers and clinicians explore these peptides in combination because their mechanisms are complementary — TB-4's stem cell activation and ILK-Akt pathway don't overlap with BPC-157's NO system modulation and VEGF upregulation. For details on combining them, see our TB-4 and BPC-157 Stack Guide.
Clinical Trial Status
TB-4's clinical development is most advanced in ophthalmology:
- RGN-259 (topical TB-4) — Phase 2 and Phase 3 clinical trials for dry eye disease and neurotrophic keratopathy. These represent the most advanced clinical testing of TB-4 in any indication (Sosne, 2018).
- Cardiac applications — Extensive preclinical evidence exists, including large animal (porcine) models (Tan et al., 2021), but human cardiac trials have not been initiated.
- Dermal wound healing — Multicenter clinical trials have been referenced in review literature (Philp & Kleinman, 2010), though published results from completed human dermal trials are limited.
The gap between extensive preclinical evidence and limited clinical data is a key limitation. While animal data is compelling across many injury types, human efficacy data is currently confined primarily to ocular applications.
Limitations of Current Research
Intellectual honesty requires acknowledging what we don't know:
Most Data Is Preclinical
The vast majority of TB-4 injury recovery research comes from rodent models. While rodent models are valuable for establishing mechanisms and proof-of-concept, they don't always predict human outcomes. Dosing, timing, and effect magnitude may differ significantly in humans.
Limited Human Data Outside Ophthalmology
Human clinical trial data for TB-4 is largely limited to corneal and ocular applications. Cardiac, neurological, dermal, and musculoskeletal applications remain preclinical despite strong animal evidence.
Dosing Protocols Are Not Standardized
Different studies use different doses, routes, and schedules. There is no consensus on optimal TB-4 dosing for any specific injury type in humans. Animal study doses require careful allometric scaling to estimate human equivalents.
Long-Term Safety Data Is Limited
While TB-4 has shown a good safety profile in animal studies and early clinical trials, long-term safety data from large human populations is not yet available.
Publication Bias
Positive results are more likely to be published than negative results. The TB-4 literature may overrepresent successful outcomes relative to the total research conducted.
Frequently Asked Questions
Can TB-4 help with my specific injury?
TB-4 has been studied in dermal wounds, corneal injuries, cardiac damage, traumatic brain injury, spinal cord injury, and burn wounds. If your injury involves tissue where cell migration, angiogenesis, and inflammation are relevant to healing (which is essentially all tissue), TB-4's mechanisms are theoretically applicable. However, direct evidence varies by injury type — the strongest data is in cardiac, corneal, and dermal applications.
How does TB-500 relate to TB-4 for injury recovery?
TB-500 is a synthetic peptide containing the active region of TB-4 responsible for actin binding, cell migration, and angiogenesis. It's widely available and used as a TB-4 analog. The core recovery mechanisms should be equivalent since TB-500 contains the same active domain, though the full-length TB-4 protein may have additional biological activities from regions outside the TB-500 fragment.
Is TB-4 better than BPC-157 for injury recovery?
Neither is universally "better." They work through different mechanisms and have different evidence bases. TB-4 has stronger evidence in cardiac, corneal, and neurological injury. BPC-157 has stronger direct evidence in tendon, ligament, and GI healing. Many researchers explore them in combination for complementary effects.
How long should TB-4 be used for injury recovery?
Research protocols typically range from 2–8 weeks depending on injury type and severity. Most animal studies show the largest gains within the first 4–6 weeks. See our TB-4 Results Timeline for a detailed week-by-week breakdown.
Is TB-4 safe?
TB-4 has shown a favorable safety profile in preclinical studies and Phase 2/3 clinical trials for ocular applications. It's a naturally occurring protein found in all nucleated human cells. However, comprehensive long-term safety data from large human populations is not yet available. Consult with a qualified healthcare provider before using any peptide.
References
- Hannappel E. beta-Thymosins. Ann N Y Acad Sci. 2007;1112:21-37. PubMed
- Sosne G, Qiu P, Christopherson PL, Wheater MK. Thymosin beta 4 suppression of corneal NFkappaB: a potential anti-inflammatory pathway. Exp Eye Res. 2007;84(4):663-669. PubMed
- Philp D, Huff T, Gho YS, et al. The actin binding site on thymosin beta4 promotes angiogenesis. FASEB J. 2003;17(14):2103-2105. PubMed
- Philp D, Nguyen M, Scheremeta B, et al. Thymosin beta4 increases hair growth by activation of hair follicle stem cells. FASEB J. 2004;18(2):385-387. PubMed
- Malinda KM, Sidhu GS, Mani H, et al. Thymosin beta4 accelerates wound healing. J Invest Dermatol. 1999;113(3):364-368. PubMed
 Thymosin Beta-4 (TB-4) is a 43-amino-acid peptide found in virtually every human cell except red blood cells. It's the body's most abundant actin-sequestering protein — a fundamental building block for cell movement, tissue repair, and wound healing. Since the late 1990s, a growing body of preclinical research has demonstrated TB-4's ability to accelerate recovery across a remarkable range of injury types.
Thymosin Beta-4 (TB-4) is a 43-amino-acid peptide found in virtually every human cell except red blood cells. It's the body's most abundant actin-sequestering protein — a fundamental building block for cell movement, tissue repair, and wound healing. Since the late 1990s, a growing body of preclinical research has demonstrated TB-4's ability to accelerate recovery across a remarkable range of injury types.
