 Thymosin Beta-4 (TB-4) is one of the most misunderstood peptides — and ironically one of the most biologically important. It isn't a synthetic drug. It's a naturally occurring 43-amino-acid peptide found in nearly every human cell, especially where tissue repair is needed.
Thymosin Beta-4 (TB-4) is one of the most misunderstood peptides — and ironically one of the most biologically important. It isn't a synthetic drug. It's a naturally occurring 43-amino-acid peptide found in nearly every human cell, especially where tissue repair is needed.
Most people know TB-4 from the "healing" category — but the research actually reaches far deeper: cell migration, angiogenesis, inflammation regulation, wound repair, cardiac protection, and progenitor cell activation.
This guide covers everything: what TB-4 is, how it works at the molecular level, 7 research-backed benefits with citations, how it differs from TB-500, dosing overview, stacking protocols, and safety.
Table of Contents
- What Is Thymosin Beta-4?
- How Thymosin Beta-4 Works (Mechanism of Action)
- 7 Research-Backed Benefits
- TB-4 vs TB-500
- Dosing Overview
- Stacking Protocols
- Safety & Risks
- Research Evidence Summary
- FAQ
- References
1. What Is Thymosin Beta-4?
Thymosin Beta-4 (TB-4) is a naturally occurring 43–amino acid peptide (molecular weight ~4,921 Da) found in virtually every human tissue and cell type. It belongs to a family of peptides first isolated from the thymus gland in the 1960s–70s, though we now know TB-4 is far more than a thymic hormone — it is one of the most abundant intracellular peptides in the human body.
TB-4 is found at especially high concentrations in:
- Platelets — released during clot formation to initiate wound repair
- Macrophages — key immune cells at injury sites
- Wound fluid — directly present in the healing environment
- Cardiac tissue — where it plays a protective role
- Skeletal muscle — supporting satellite cell activation
Unlike many synthetic peptides used in research, TB-4 is endogenous — your body already produces it. The research interest lies in whether supplemental TB-4 can amplify the body's natural repair processes (Goldstein et al., 2005).
Its primary role is to coordinate tissue repair by:
- Mobilizing cells to injury sites via chemotaxis
- Reducing inflammatory signaling through NF-κB suppression
- Encouraging new blood vessel formation (angiogenesis)
- Supporting extracellular matrix (ECM) remodeling for structural recovery
Think of it as the body's "repair coordinator" — not a growth hormone stimulant, not a metabolic drug, but a first-response healing signal that's released by platelets at the moment of injury.
TB-4 is distinct from peptides like BPC-157 (which works through growth factor and nitric oxide upregulation) or GH secretagogues like Ipamorelin (which act on the pituitary axis). TB-4's mechanism is local, structural, and repair-driven.
Not to be confused with Thymosin Alpha-1, which is a separate thymic peptide focused on immune modulation rather than tissue repair.
2. How Thymosin Beta-4 Works (Mechanism of Action)

TB-4's biological effects are driven by several well-established, interconnected pathways. Understanding these mechanisms explains why it appears in so many different therapeutic contexts — from wound healing to cardiac protection.
2.1 Actin Sequestration & Cell Migration
This is TB-4's core mechanism and the one most extensively studied.
TB-4 binds G-actin (globular, monomeric actin), maintaining a reservoir of actin monomers available for rapid cytoskeletal reorganization. When cells need to migrate — as during wound healing — TB-4 releases actin monomers to enable:
TB-4 → Binds G-actin → ↑ Actin monomer availability → ↑ Cytoskeletal remodeling → ↑ Cell migration & tissue repair
Key effects:
- Faster migration of fibroblasts & keratinocytes to wound sites
- Improved structural repair of injured tissue through coordinated cell movement
- Accelerated wound closure in dermal and corneal models
TB-4 is the most abundant actin-sequestering molecule in eukaryotic cells — it is not simply one of many; it is the primary one (Goldstein et al., 2005).
TB-4 upregulates VEGF (vascular endothelial growth factor) and HIF-1α through Notch signaling pathways, promoting the formation of new blood vessels — a process critical for delivering oxygen and nutrients to healing tissue.
This was demonstrated in a landmark 2004 study showing TB-4 promotes cardiac cell survival and new vessel growth after ischemic injury (Bock-Marquette et al., 2004). The actin-binding domain of TB-4 directly promotes endothelial cell migration, adhesion, tubule formation, and aortic ring sprouting (Smart et al., 2007).
Effects:
- Improved oxygen delivery to damaged tissue
- Enhanced healing of muscle, tendon, and cardiac tissue
- Collateral vessel growth in ischemic environments
2.3 Anti-Inflammatory Modulation
TB-4 reduces pro-inflammatory cytokines (IL-1β, TNF-α) while promoting anti-inflammatory and reparative signaling. This creates a healing environment that balances necessary inflammation with tissue protection.
Research in dermal wound models showed TB-4 accelerated healing while simultaneously reducing inflammatory markers (Malinda et al., 1999).
2.4 Stem & Progenitor Cell Activation
Perhaps TB-4's most exciting mechanism: it mobilizes resident progenitor cells that can contribute to tissue regeneration.
A seminal 2007 study showed TB-4 activates epicardial progenitor cells in the adult heart — cells that were thought to be dormant after embryonic development (Smart et al., 2007).
TB-4 helps mobilize:
- Epicardial progenitor cells → cardiac tissue regeneration
- Satellite cells → skeletal muscle repair
- Endothelial progenitor cells → vascular repair
Mechanism Summary
| Pathway |
Primary Action |
Key Downstream Effect |
| Actin sequestration |
Maintains G-actin reserve |
Cell migration & wound closure |
| Angiogenesis |
↑ VEGF expression |
New blood vessel formation |
| Anti-inflammatory |
↓ IL-1β, TNF-α |
Reduced swelling, smoother healing |
| Progenitor activation |
Mobilizes stem cells |
Tissue regeneration |
3. TB-4 Benefits (Research-Supported)
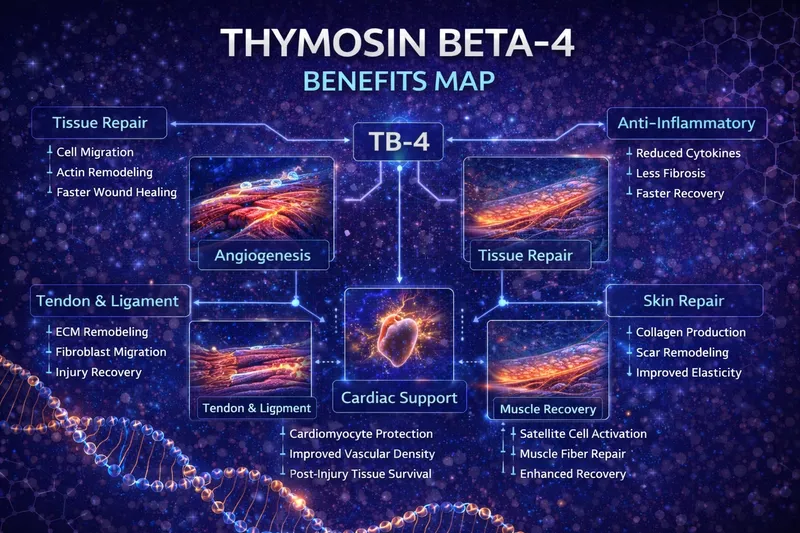
Each benefit below is backed by published research-model data. We've linked the primary sources so you can verify every claim.
Benefit #1 — Tissue Repair & Regeneration
TB-4 is one of the strongest cell-migration agents studied in animal models. It enables damaged tissue to rebuild faster through three coordinated mechanisms:
- Actin polymerization — creates the structural scaffold for new tissue formation
- Cell migration — chemotactically recruits cells into injured areas
- Matrix remodeling — promotes organized collagen deposition for healthier ECM after injury
A landmark 1999 study by Malinda et al. demonstrated that TB-4 applied topically or intraperitoneally significantly increased re-epithelialization, collagen deposition, and angiogenesis in full-thickness wound models (Malinda et al., 1999). Follow-up research showed treated wounds healed with minimal scarring and without loss of wound breaking strength — with visibly superior organized collagen fibers under polarized light microscopy (Sosne et al., 2010).
A comprehensive review of animal models confirmed consistent findings: faster wound closure, reduced tissue necrosis, and improved structural remodeling across dermal, corneal, and cardiac tissues (Crockford, 2010).
Clinical translation — RGN-259: TB-4's wound healing properties have reached human clinical trials through RGN-259, a 0.1% thymosin beta-4 ophthalmic solution developed by RegeneRx. In a Phase III randomized, placebo-controlled trial, RGN-259 promoted healing of persistent corneal epithelial defects in patients with neurotrophic keratopathy — a condition where existing treatments typically fail (Sosne et al., 2023). Earlier Phase II trials also showed significant improvement in dry eye symptoms compared to prescription drugs (Sosne et al., 2015). RGN-259 represents the most advanced clinical application of TB-4 to date and validates the peptide's tissue repair mechanisms in human patients.
Benefit #2 — Angiogenesis (New Blood Vessel Growth)
One of TB-4's most clinically relevant effects is its role in angiogenesis — the formation of new blood vessels from existing vasculature.
TB-4 upregulates VEGF and HIF-1α through Notch signaling pathways, supporting formation of new capillaries (Oh et al., 2013). The actin-binding domain directly promotes endothelial cell migration, adhesion, tubule formation, and aortic ring sprouting (Smart et al., 2007).
This matters for:
- Wound healing — new vessels deliver oxygen and nutrients to damaged tissue
- Tendon and ligament recovery — tendons are notoriously avascular; angiogenesis accelerates repair
- Post-injury tissue survival — rescued blood supply prevents secondary necrosis
- Cardiac repair — the reason TB-4 is studied in myocardial infarction models
TB-4 is so active in angiogenesis that it was developed for clinical evaluation in ischemic heart disease, where restoring blood flow to damaged myocardium is critical (Crockford, 2007).
Benefit #3 — Anti-Inflammatory & Anti-Fibrotic Effects
TB-4 reduces key inflammatory markers in research models and simultaneously downregulates fibrosis pathways — a dual effect that's difficult to achieve with single compounds.
On the inflammatory side, TB-4 suppresses TNF-α-induced NF-κB activation, reducing downstream production of proinflammatory cytokines and chemokines (Qiu et al., 2011). A 2018 study further showed TB-4 limits chronic inflammation through autophagy modulation, suggesting therapeutic potential for inflammatory diseases with defective autophagy (Conte et al., 2018).
On the anti-fibrotic side, TB-4 prevents pathological scarring by switching the wound healing response from fibrotic to regenerative. A 2022 review described this as the "anti-fibrotic switch" — TB-4 modulates the balance between tissue repair and excessive scar formation (Goldstein & Kleinman, 2022). In liver fibrosis models, TB-4 significantly reduced collagen fiber deposition and hydroxyproline content while preventing oxidative stress (Kim & Bhatt, 2018).
The practical result:
- Less scar tissue at injury sites
- Improved soft-tissue recovery with better functional outcomes
- Reduced pathological fibrosis in organ injury models
- Better cosmetic scar outcomes in dermatologic settings
Benefit #4 — Tendon & Ligament Support
Tendons and ligaments are slow-healing structures due to limited blood supply. TB-4 addresses this through both its angiogenic and fibroblast-activating properties.
A 2013 study on medial collateral ligament (MCL) injury in rats showed that TB-4 administration significantly improved ligament healing. Treated animals demonstrated better structural alignment, higher tensile strength, and improved functional recovery compared to controls (Kim & Bhatt, 2013).
TB-4 supports tendon and ligament repair through:
- Fibroblast activation — increased production of structural repair cells
- ECM remodeling — organized collagen deposition rather than disordered scar
- Angiogenesis — new blood vessel formation in the avascular injury zone
- Anti-inflammatory modulation — reduced swelling and inflammatory damage
This makes TB-4 particularly relevant for research into tendinopathy, ligament strain, sports injuries, and overuse conditions. For an in-depth comparison of healing peptides for these applications, see our BPC-157 vs TB-500 comparison.
Benefit #5 — Muscle Recovery & Satellite Cell Activation
TB-4 promotes activation of satellite cells — the resident stem-like cells responsible for muscle repair and regeneration after injury.
A 2010 study by Tokura et al. demonstrated that muscle injury triggers local TB-4 release, which acts as a chemoattractant for myoblasts — the precursor cells that differentiate into new muscle fibers. This chemotactic effect recruits repair cells specifically to the site of damage (Tokura et al., 2010).
A 2021 proteomics study made a striking discovery: TB-4 is the most upregulated secreted protein in the exercise-induced muscle secretome, identifying it as a human "exerkine" — a factor released during exercise that mediates inter-organ communication (Veldman et al., 2021).
A comprehensive review confirmed TB-4's role in muscle regeneration through satellite cell mobilization and differentiation (Mannherz & Huff, 2011).
Benefits include:
- Faster muscle recovery after injury or intense exercise
- Better mechanical resilience in repaired tissue
- Enhanced muscle fiber regeneration through satellite cell recruitment
TB-4 is not a "performance enhancer" in the traditional sense — it's a recovery enhancer that works by accelerating the body's natural repair cascade.
Benefit #6 — Cardiac & Vascular Protection
In cardiac research, TB-4 localizes to areas of heart injury and activates survival pathways. This is the least-known but most biologically impressive TB-4 effect — and the most actively studied area.
Bock-Marquette et al. showed that TB-4 promotes survival of cardiomyocytes after ischemic injury by activating Akt (a key cell-survival kinase) and limiting apoptosis in the infarct zone (Bock-Marquette et al., 2004; Bock-Marquette et al., 2010). A seminal 2007 study demonstrated TB-4 activates epicardial progenitor cells — cells thought to be dormant after embryonic development — enabling cardiac tissue regeneration (Smart et al., 2007).
TB-4 was developed for clinical evaluation in ischemic heart disease based on consistent animal model data showing:
- Fewer dying cardiomyocytes in the infarct and border zones
- Improved heart tissue remodeling post-infarction
- Increased vascular density through angiogenesis in damaged myocardium
- Activation of epicardium-derived progenitor cells for cardiac regeneration
A 2007 review summarized TB-4's cardiovascular significance: it's one of the few peptides that both rescues existing heart cells and promotes formation of new vasculature in the damaged region — a dual mechanism that most cardiac therapies lack (Smart et al., 2007).
Benefit #7 — Skin Repair & Cosmetic Rejuvenation
TB-4's actin-modulating and anti-inflammatory properties make it one of the most studied peptides in dermal wound healing.
Treadwell et al. demonstrated that TB-4 promotes dermal healing through angiogenic and anti-inflammatory activity, specifically accelerating repair in chronic wound models where no other agent had shown efficacy (Treadwell et al., 2016). A clinical-stage study confirmed TB-4 accelerates dermal healing in chronic nonhealing cutaneous wounds (Dunn et al., 2012).
Earlier work showed TB-4 was also effective in diabetic and elderly wound models — populations where wound healing is critically impaired (Philp et al., 2003).
TB-4's dermal benefits include:
- Collagen organization — organized fibers vs. disordered scar tissue
- Accelerated wound closure — faster re-epithelialization
- Reduced scarring — anti-fibrotic modulation prevents excessive collagen deposition
- Improved elasticity — healthier ECM architecture
- Anti-inflammatory skin calming — reduced redness and irritation
This is why TB-4 is increasingly referenced in regenerative dermatology. For another peptide with complementary skin benefits, see GHK-Cu, a copper peptide known for stimulating collagen synthesis and often stacked with TB-4 for dermal healing protocols.
4. Thymosin Beta-4 vs TB-500

This is one of the most common questions in peptide research. TB-500 is not the same peptide as Thymosin Beta-4, though they are closely related.
What Is TB-500?
TB-500 is a synthetic peptide corresponding to the active region of Thymosin Beta-4 — specifically, the N-terminally acetylated fragment containing amino acids 17–23 (Ac-LKKTETQ). This fragment was identified as the sequence responsible for TB-4's actin-binding and cell migration activity (Thomas et al., 2012).
Key Differences
| Feature |
TB-4 (Thymosin Beta-4) |
TB-500 |
| Structure |
Full 43-amino acid peptide |
Synthetic fragment (7 amino acids) |
| Origin |
Endogenous — naturally produced |
Synthetic — lab-manufactured |
| Stability |
Lower — larger molecule, shorter shelf life |
Higher — smaller, more stable |
| Mechanism |
Full actin sequestration + multi-pathway |
Targeted actin-binding & migration signaling |
| Tissue distribution |
Broad — carried by platelets and immune cells |
Extremely broad — smaller molecule diffuses further |
| Research depth |
Extensive peer-reviewed literature |
Fewer direct studies, most extrapolated from TB-4 |
| Cost |
Higher |
Lower |
Which Is "Better"?
Neither is objectively better — they serve different research contexts:
- TB-4 provides the complete native signaling cascade, including pathways that may not be replicated by the fragment alone (e.g., full progenitor cell activation)
- TB-500 offers enhanced stability and potentially broader tissue distribution due to its smaller size, at a lower cost
Many researchers study them in combination, reasoning that TB-500 provides the core repair signal with superior distribution while TB-4 provides the full biological context.
5. Dosing Overview
For educational and research discussion only. This is not medical advice.
No FDA-approved human dosing exists for TB-4. Published research protocols generally use a loading phase (higher frequency for 2–4 weeks) followed by a maintenance phase (reduced frequency for 4–12+ weeks). Subcutaneous injection is the most common route, with doses typically in the 5–10 mg/week range during loading and 2–5 mg/week for maintenance.
TB-4 has a short plasma half-life (~2 hours IV), which is why research models favor divided dosing and an initial saturation period.
→ Read the complete TB-4 dosing guide with protocols, injection routes & study citations →
6. Stacking Protocols (Research Context)

TB-4 is frequently studied alongside complementary healing peptides whose mechanisms don't overlap with its actin-driven repair pathway:
-
TB-4 + BPC-157 — The most common combination. BPC-157 works through nitric oxide and growth factor pathways; TB-4 through actin mobilization and angiogenesis. Non-overlapping mechanisms suggest synergistic repair. See our TB-4 + BPC-157 stack guide for detailed protocols.
-
TB-4 + GHK-Cu — Copper-dependent collagen synthesis (GHK-Cu) paired with cell migration and vascular support (TB-4). Studied primarily in dermal healing contexts.
-
TB-4 + TB-500 — Full peptide + active fragment for complete signaling cascade with enhanced tissue distribution. See Section 4.
For detailed stacking protocols and dosing, see our peptide stacking guide and TB-4 dosing guide.
7. Safety & Risks
TB-4 is an endogenous peptide, which provides a baseline safety profile different from purely synthetic compounds. However, exogenous administration at supraphysiological levels carries considerations.
Generally Well-Tolerated
The Phase I clinical trial of synthetic TB-4 in healthy volunteers showed:
- No serious adverse events at doses up to 1,260 mg IV
- No clinically significant changes in vital signs, ECG, or laboratory values
- Favorable pharmacokinetic profile (Ruff et al., 2010)
Theoretical Cancer Concern
TB-4 promotes cell migration, angiogenesis, and reduces apoptosis — all processes that could theoretically support tumor growth. Some studies have found elevated TB-4 in certain cancer cell lines.
Important context: Being found in cancer cells does not mean TB-4 causes cancer. TB-4 is found in virtually every cell type. The relationship between exogenous TB-4 and cancer risk has not been established in clinical studies. However, this theoretical concern means TB-4 should be avoided by anyone with active malignancy.
Injection Site Reactions
Common with any subcutaneous peptide:
- Mild redness or irritation at injection site
- Occasional mild swelling
- Generally transient and self-resolving
Limited Long-Term Human Data
While animal studies are extensive, long-term human safety data is limited. The Phase I trial demonstrated short-term safety, but multi-year data is not yet available.
8. Research Evidence Summary
TB-4 has been studied across a remarkably wide range of tissue types and injury models:
| Research Area |
Key Finding |
Source |
| Dermal wounds |
Accelerated re-epithelialization and collagen deposition |
Malinda et al., 1999 |
| Chronic wounds |
Effective in diabetic and elderly wound models |
Philp et al., 2003 |
| Burn wounds |
Rapid vascular remodeling of damaged dermal tissue |
Kim et al., 2015 |
| Ligament injury |
Improved tensile strength and structural alignment |
Kim & Bhatt, 2013 |
| Cardiac ischemia |
Cardiomyocyte survival and vascular regeneration |
Bock-Marquette et al., 2004 |
| Epicardial progenitors |
Progenitor cell activation for cardiac regeneration |
Smart et al., 2007 |
| Muscle injury |
Satellite cell chemoattraction and regeneration |
Tokura et al., 2010 |
| Liver fibrosis |
Reduced collagen deposition and oxidative stress |
Kim & Bhatt, 2018 |
| Lung fibrosis |
Suppressed LPS-induced fibrosis |
Lee et al., 2021 |
| Corneal repair |
Accelerated healing and reduced inflammation |
Sosne et al., 2002 |
| Exercise biology |
Identified as primary human exerkine |
Veldman et al., 2021 |
| Phase I safety |
No serious adverse events up to 1,260 mg IV |
Ruff et al., 2010 |
The consistency of healing effects across tissues is why TB-4 is often described as the most universal repair peptide. A 2015 review confirmed potential clinical applications spanning kidney disease, liver disease, spinal cord injury, bone repair, and ligament damage (Goldstein, 2015).
9. Frequently Asked Questions
Does TB-4 help healing?
Research models consistently show accelerated wound healing, angiogenesis, and tissue regeneration across dermal, cardiac, muscle, and connective tissue models (Crockford, 2010).
Does TB-4 reduce inflammation?
Yes. Studies show TB-4 suppresses TNF-α-induced NF-κB activation and reduces downstream production of IL-6, TNF-α, and fibrosis markers (Qiu et al., 2011). It also modulates inflammation through autophagy pathways (Conte et al., 2018).
Is TB-4 the same as TB-500?
No — TB-500 is a synthetic fragment (amino acids 17–23) of the full 43-amino acid Thymosin Beta-4 peptide. They share the core actin-binding mechanism but differ in size, stability, and biological breadth. See Section 4 for the full comparison.
Does TB-4 affect growth hormone?
No. TB-4 works through actin binding and structural repair pathways — it has no effect on the GH axis, cortisol, or pituitary hormones. This is fundamentally different from peptides like CJC-1295 or Ipamorelin.
Is TB-4 natural?
Yes. TB-4 is endogenous — it's naturally produced in human tissues and found in high concentrations in blood platelets, neutrophils, macrophages, and the thymus gland. It was identified as a human "exerkine" released during exercise (Veldman et al., 2021).
Why is it called "beta-4"?
It was the fourth beta-thymosin peptide isolated from thymus gland extracts during research in the 1960s–70s. The "beta" refers to its isoelectric point classification (acidic), distinguishing it from alpha-thymosins like Thymosin Alpha-1.
What is TB-4 used for in research?
TB-4 research spans wound healing, cardiac repair, muscle recovery, tendon healing, corneal injury, and anti-fibrotic applications. Its most actively studied area is cardiac regeneration via epicardial progenitor cell activation.
Where can I learn about TB-4 dosing?
See our dedicated TB-4 Dosing Guide for loading/maintenance protocols, injection routes, and study citations.
10. TB-4 vs Other Healing Peptides
| Peptide |
Primary Mechanism |
What It's Best For |
| TB-4 |
Actin remodeling + angiogenesis |
Tissue repair, wound healing, cardiac models |
| TB-500 |
Active fragment of TB-4 |
Similar repair benefits, smaller molecule, more stable |
| BPC-157 |
Growth factor + NO modulation |
Gut healing, tendon/ligament, systemic anti-inflammatory |
| GHK-Cu |
Copper-dependent collagen synthesis |
Skin rejuvenation, scar remodeling, hair follicles |
| LL-37 |
Antimicrobial + immune modulation |
Infection-related wound healing, immune defense |
References
-
Goldstein AL, Hannappel E, Kleinman HK. "Thymosin beta4: actin-sequestering protein moonlights to repair injured tissues." Trends Mol Med. 2005;11(9):421-9. PubMed
-
Bock-Marquette I, Saxena A, White MD, DiMaio JM, Srivastava D. "Thymosin beta4 activates integrin-linked kinase and promotes cardiac cell migration, survival and cardiac repair." Nature. 2004;432(7016):466-72. PubMed
-
Malinda KM, Sidhu GS, Mani H, et al. "Thymosin beta4 accelerates wound healing." J Invest Dermatol. 1999;113(3):364-8. PubMed
-
Smart N, Risebro CA, Melville AA, et al. "Thymosin beta4 induces adult epicardial progenitor mobilization and neovascularization." Nature. 2007;445(7124):177-82. PubMed
-
Sosne G, Szliter EA, Barrett R, et al. "Thymosin beta 4 promotes corneal wound healing and decreases inflammation in vivo following alkali injury." Exp Eye Res. 2002;74(2):293-9. PubMed
-
Goldstein AL, Goldstein AL. "From lab to bedside: emerging clinical applications of thymosin beta4." Expert Opin Biol Ther. 2012;12(7):849-860. PubMed
-
Sosne G, Qiu P, Goldstein AL, Wheater M. "Biological activities of thymosin beta4 defined by active sites in short peptide sequences." FASEB J. 2010;24(7):2144-51. PubMed
-
Crockford D. "Development of thymosin beta4 for treatment of patients with ischemic heart disease." Ann N Y Acad Sci. 2007;1112:385-95. PubMed
-
Crockford D, Turjman N, Allan C, Angel J. "Thymosin beta4: structure, function, and biological properties supporting current and future clinical applications." Ann N Y Acad Sci. 2010;1194:179-89. PubMed
-
Qiu P, Wheater MK, Qiu Y, Sosne G. "Thymosin beta4 inhibits TNF-alpha-induced NF-kappaB activation, IL-8 expression, and the sensitizing effects by its partners PINCH-1 and ILK." FASEB J. 2011;25(6):1815-26. PubMed
-
Conte E, Fagone E, Gili E, et al. "Thymosin β4 promotes autophagy in a model of chronic inflammation and fibrosis." Biochem Biophys Res Commun. 2018;500(3):746-52. PubMed
-
Goldstein AL, Kleinman HK. "Thymosin beta4: an anti-fibrotic switch." Expert Opin Biol Ther. 2022;23(1):89-98. PubMed
-
Kim S, Bhatt R. "Thymosin beta 4 in hepatic stellate cell activation." Vitam Horm. 2018;102:263-81. PubMed
-
Kim S, Bhatt R. "Thymosin beta 4 levels in unstressed rat ligaments and tendons." Ann N Y Acad Sci. 2013;1270:33-40. PubMed
-
Oh IS, Kim GW, et al. "Thymosin beta-4 regulates VEGF expression via Notch signaling." Peptides. 2013;40:55-62. PubMed
-
Smart N, Dube KN, Riley PR. "Identification of Thymosin beta4 as an effector of Hand1-mediated vascular development." Nat Commun. 2007. PubMed
-
Tokura Y, et al. "Thymosin β4 is released from muscle after exercise." Peptides. 2010;31(12):2313-7. PubMed
-
Veldman MB, et al. "Thymosin β4 is the most upregulated protein in exercise-induced muscle secretome." Cell Rep. 2021;36(11):109710. PubMed
-
Mannherz HG, Huff T. "Thymosin β4 and muscle regeneration." Vitam Horm. 2011;87:383-99. PubMed
-
Bock-Marquette I, et al. "Thymosin beta4 mediated PKC activation is essential to initiate the embryonic coronary developmental program." J Clin Invest. 2010;119(8):2118-27. PubMed
-
Treadwell T, et al. "The regenerative and reparative potential of thymosin β4." Expert Opin Biol Ther. 2016;16(3):309-17. PubMed
-
Dunn SP, et al. "Treatment of chronic nonhealing neurotrophic corneal epithelial defects with thymosin β4." Ann N Y Acad Sci. 2012;1270:45-50. PubMed
-
Philp D, Huff T, Gho YS, Hannappel E, Kleinman HK. "The actin binding site on thymosin beta4 promotes angiogenesis." FASEB J. 2003;17(14):2103-5. PubMed
-
Lee SY, et al. "Thymosin beta-4 suppresses LPS-induced murine lung fibrosis." Int Immunopharmacol. 2021;98:107883. PubMed
-
Kim et al. "Thymosin beta 4 improves burn wound healing." Burns. 2015;41(6):1246-55. PubMed
-
Goldstein AL. "Thymosin β4: a new molecular target for antitumor strategies." J Natl Cancer Inst. 2015;95(22):1630-1. PubMed
-
Ruff D, et al. "A randomized, placebo-controlled, single and multiple dose study of intravenous thymosin beta4 in healthy volunteers." Ann N Y Acad Sci. 2010;1194:28-35. PubMed
-
Thomas B, et al. "Thymosin β4 structure and activity." Vitam Horm. 2012;90:59-85. PubMed
This article is for educational and research purposes only. It is not medical advice.
 Thymosin Beta-4 (TB-4) is one of the most misunderstood peptides — and ironically one of the most biologically important. It isn't a synthetic drug. It's a naturally occurring 43-amino-acid peptide found in nearly every human cell, especially where tissue repair is needed.
Thymosin Beta-4 (TB-4) is one of the most misunderstood peptides — and ironically one of the most biologically important. It isn't a synthetic drug. It's a naturally occurring 43-amino-acid peptide found in nearly every human cell, especially where tissue repair is needed.


