Something is wrong with your digestion and you cannot pin down what. Food intolerances that developed out of nowhere. Bloating after meals that used to sit fine. Fatigue that does not respond to more sleep. Maybe a doctor mentioned "increased intestinal permeability" or you have read about "leaky gut" and wondered if that is what is happening.
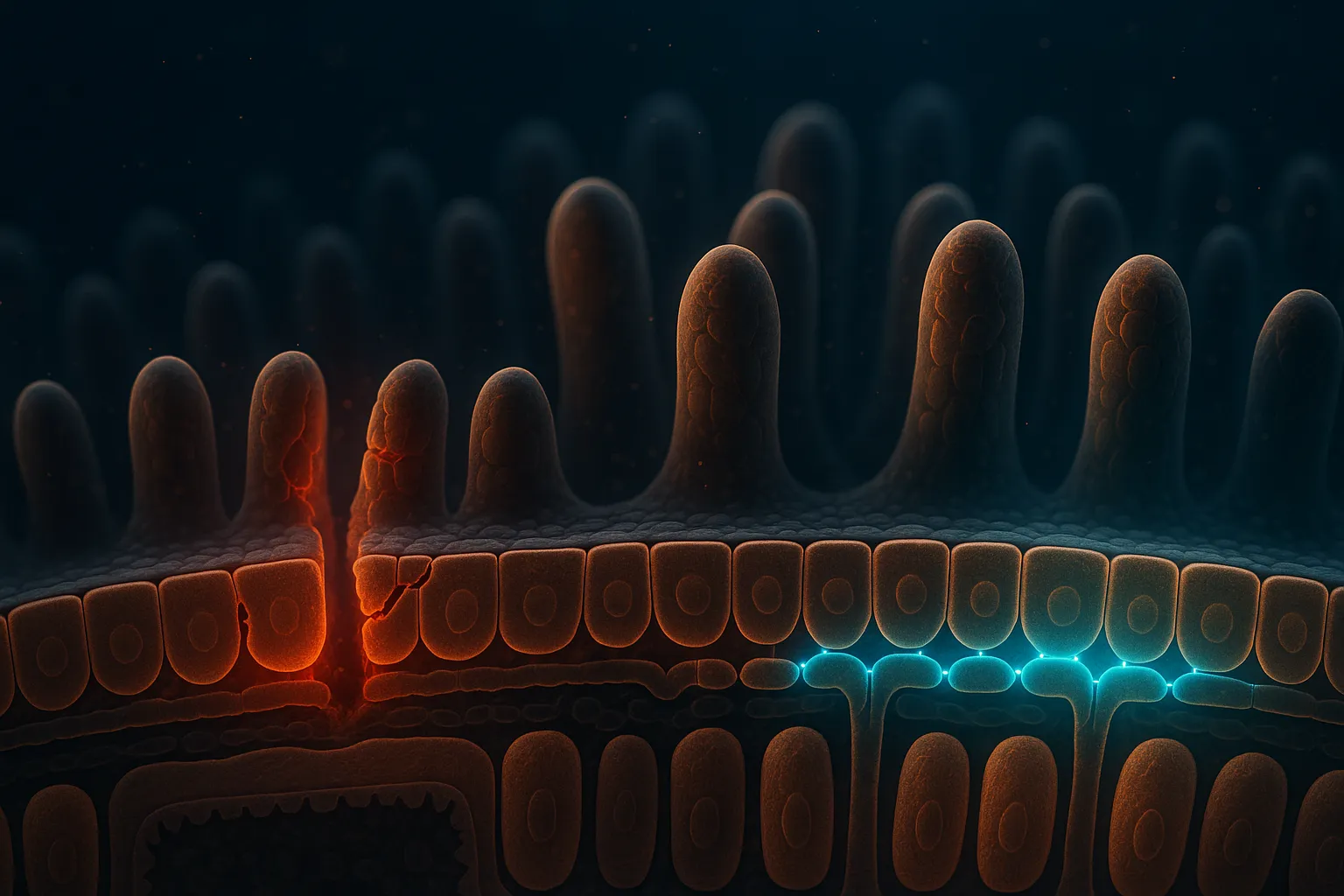
Here is what is actually going on at the biological level: your intestinal barrier is a single-cell-thick layer that separates the inside of your gut (and everything you eat) from your bloodstream and immune system. These cells are held together by tight junction proteins — molecular gates that control what passes through. When these tight junctions weaken, molecules that should stay in the gut (food particles, bacterial toxins, inflammatory compounds) leak into the bloodstream and trigger immune responses. The result is systemic inflammation, food sensitivities, brain fog, and digestive symptoms that seem disconnected from what you ate.
The challenge is that most conventional approaches support the ecosystem around the barrier — the microbiome, the mucus layer, the immune cells — without directly targeting the barrier cells and tight junctions themselves. Understanding this distinction is key to finding approaches that actually repair the lining rather than just managing symptoms.
Conventional Approaches: What Helps and Where the Gap Is
Elimination diets are the essential first step. You cannot heal a barrier while continuing to expose it to triggers. The most common culprits — gluten, dairy, processed seed oils, alcohol, and high-FODMAP foods — should be systematically removed and reintroduced. This does not heal the lining, but it stops the ongoing damage that prevents healing.
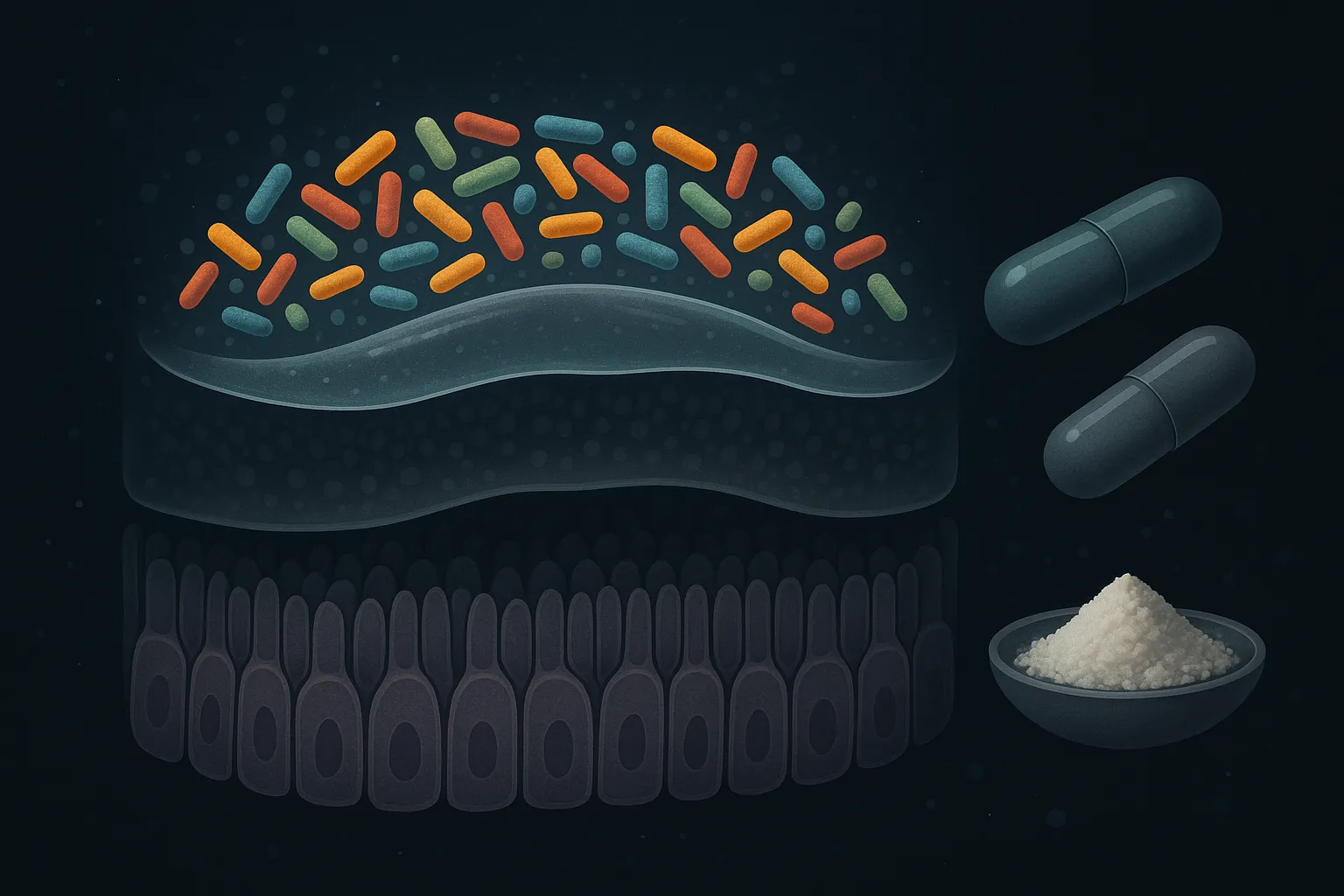
Probiotics support the microbial ecosystem on top of the gut lining. Specific strains (Lactobacillus rhamnosus GG, Saccharomyces boulardii, Bifidobacterium infantis) have evidence for reducing inflammation and supporting barrier function indirectly. But probiotics do not repair damaged epithelial cells or restore weakened tight junctions — they maintain the environment around the barrier.
L-glutamine is the most-cited gut repair supplement, and it has legitimate evidence. Glutamine is the primary fuel source for intestinal epithelial cells (enterocytes), and supplementation has been shown to reduce intestinal permeability in both animal models and clinical studies (PMID:27749689). A systematic review confirmed significant permeability reduction with glutamine supplementation above 30g/day (PMID:39397201). It is inexpensive, safe, and a reasonable baseline supplement for gut repair.
Bone broth and collagen provide amino acids (glycine, proline, hydroxyproline) that are building blocks for connective tissue. The traditional use for gut healing has some supporting data — collagen peptides may support intestinal barrier integrity — but the effect size is modest and the evidence is mostly mechanistic rather than from controlled trials.
Zinc carnosine has good evidence specifically for gastric (stomach) lining repair. It chelates to the stomach wall and promotes mucosal healing. For stomach-specific issues like gastritis or NSAID damage, zinc carnosine is well-supported. Its effects on intestinal (lower gut) permeability are less established.
Anti-inflammatory diet (Mediterranean, AIP, or low-inflammatory approaches) reduces the immune burden on the gut and creates conditions for healing. This is helpful but slow — dietary changes alone may take months to produce measurable barrier improvement, and they depend on the damage not being too severe.
The gap: the intestinal barrier is a single-cell-thick layer with tight junction proteins that require specific molecular signals to regenerate and strengthen. L-glutamine fuels the cells. Probiotics maintain the environment. Elimination diets remove triggers. But few conventional supplements directly signal the epithelial cells to proliferate, restore tight junctions, or resolve the inflammatory cascades that perpetuate barrier dysfunction at the cellular level.
The Research Frontier: Compounds That Target the Barrier Directly
A class of research compounds — peptides — approaches gut healing differently. Rather than supporting the environment around the barrier, these compounds target the biological machinery of barrier repair itself: mucosal regeneration, inflammatory pathway inhibition, and antimicrobial defense.
Peptides are short chains of amino acids that act as signaling molecules in the body. For gut health, three peptides have distinct and complementary mechanisms — one promotes mucosal healing and angiogenesis, one inhibits the master inflammatory switch (NF-kB) in gut tissue, and one targets pathogenic bacteria while supporting mucosal immunity.
What makes gut-specific peptides particularly interesting is that one of them — BPC-157 — is naturally derived from human gastric juice and can be taken orally, providing direct contact with the intestinal lining. This is unusual among peptides, which typically require injection.
For a foundational understanding of what peptides are and how they work, see What Are Peptides?.
How Gut Peptides Work Differently Than Supplements
Conventional gut supplements work through nutritional support — providing fuel (glutamine), building blocks (collagen amino acids), or ecological management (probiotics). Peptides work through signaling — they bind to specific receptors on intestinal cells and trigger repair cascades.
BPC-157 upregulates VEGF (vascular endothelial growth factor) at the gut mucosal level, promoting new blood vessel formation that delivers oxygen and nutrients to damaged tissue. It also modulates the nitric oxide system, which regulates blood flow, inflammation, and cellular protection in the GI tract. KPV directly inhibits NF-kB — the master transcription factor that drives inflammatory gene expression in gut tissue. LL-37 provides antimicrobial defense, selectively targeting pathogenic bacteria while preserving beneficial microbiota.
The analogy: if your gut lining is a damaged wall, glutamine provides bricks, collagen provides mortar, probiotics maintain the garden around the wall, and gut peptides send the construction crew with specific instructions about what to rebuild and how.
The Key Peptides for Gut Repair
BPC-157 — The Gut's Native Repair Signal
BPC-157 (Body Protection Compound) is a 15-amino-acid peptide isolated from human gastric juice. It is not a foreign molecule — it is derived from a protein your stomach naturally produces. This origin explains its unique oral stability and its affinity for gastrointestinal tissue.
The gut-specific evidence is extensive. BPC-157 has been studied in models of ulcerative colitis, gastric ulcers, intestinal anastomosis healing, NSAID-induced gut damage, and short bowel syndrome. A comprehensive review demonstrated its effectiveness across the entire gastrointestinal tract, noting its cytoprotective and healing properties from esophagus to colon (PMID:21548867). Focused research on ulcerative colitis models showed significant reduction in mucosal damage and inflammatory markers (PMID:22300085).
BPC-157 has entered phase 2 clinical trials for inflammatory bowel disease (trial designations PL-10, PLD-116, PL14736), making it one of the few research peptides with a clinical development pathway for GI applications (PMID:17713731).
For gut-specific use, BPC-157 is typically taken orally — providing direct contact with the intestinal mucosa. This is the only healing peptide with strong evidence for oral bioavailability and gut-specific effects.
For a complete overview of all BPC-157 applications, see BPC-157 Benefits.
Top BPC-157 Vendors
Ranked by price, COA availability, and reputation
KPV — Targeted Inflammatory Shutdown
KPV is a tripeptide (Lys-Pro-Val) derived from alpha-melanocyte-stimulating hormone (alpha-MSH). Its mechanism is precise: KPV inhibits the activation of NF-kB — the master transcription factor that drives inflammatory gene expression throughout the body, including the gut mucosa.
In murine colitis models, KPV significantly reduced intestinal inflammation in both DSS-induced and TNBS-induced colitis, decreasing pro-inflammatory cytokine expression (PMID:18092346). The mechanism involves PepT1-mediated uptake — KPV enters intestinal epithelial cells through the PepT1 transporter and inhibits NF-kB and MAP kinase signaling at nanomolar concentrations (PMID:18061177).
KPV is particularly relevant for inflammatory bowel conditions (Crohn's, ulcerative colitis) or any gut dysfunction driven primarily by chronic inflammation. Where BPC-157 promotes regeneration, KPV silences the inflammatory signals that prevent regeneration from occurring.
Top KPV Vendors
Ranked by price, COA availability, and reputation
LL-37 — Antimicrobial Barrier Defense
LL-37 is the only human cathelicidin — an antimicrobial peptide that your body naturally produces as part of innate immune defense. It is expressed in the intestinal epithelium and plays a critical role in maintaining the balance between beneficial and pathogenic gut bacteria.
Research shows LL-37 preserves intestinal barrier function by protecting goblet cells, increasing transepithelial resistance, and upregulating mucin-2 expression (PMID:36958193). It also contributes to maintenance and re-establishment of intestinal barrier integrity through both direct antimicrobial action and indirect immune modulation (PMID:19328825).
LL-37 is most relevant for gut dysfunction involving microbial imbalance — SIBO (small intestinal bacterial overgrowth), post-antibiotic dysbiosis, or recurrent gut infections. It selectively targets pathogenic bacteria while preserving beneficial species, which broad-spectrum antibiotics cannot do.
Top LL-37 Vendors
Ranked by price, COA availability, and reputation
For a full comparison of all gut health peptides, including protocols and stacking guidance, see Best Peptides for Gut Health.
