The Wolverine Stack — BPC-157 + TB-500 — has one of the more favorable safety profiles among peptide stacks, based on the available data. BPC-157 has been through human clinical trials without significant adverse events. TB-500 has not.
That asymmetry matters. BPC-157's safety data comes from controlled trials. TB-500's safety profile comes from animal studies and community self-reports. The combination has never been formally tested in humans.
Most users report mild, transient effects that resolve within days. But there's one theoretical concern — TB-500 and angiogenesis-related cancer risk — that deserves honest discussion rather than dismissal.
For dosing and protocols, see the Wolverine Stack Dosing Guide. For benefits and mechanism, see the Wolverine Stack Benefits guide.
Common Side Effects (>10% of Users)
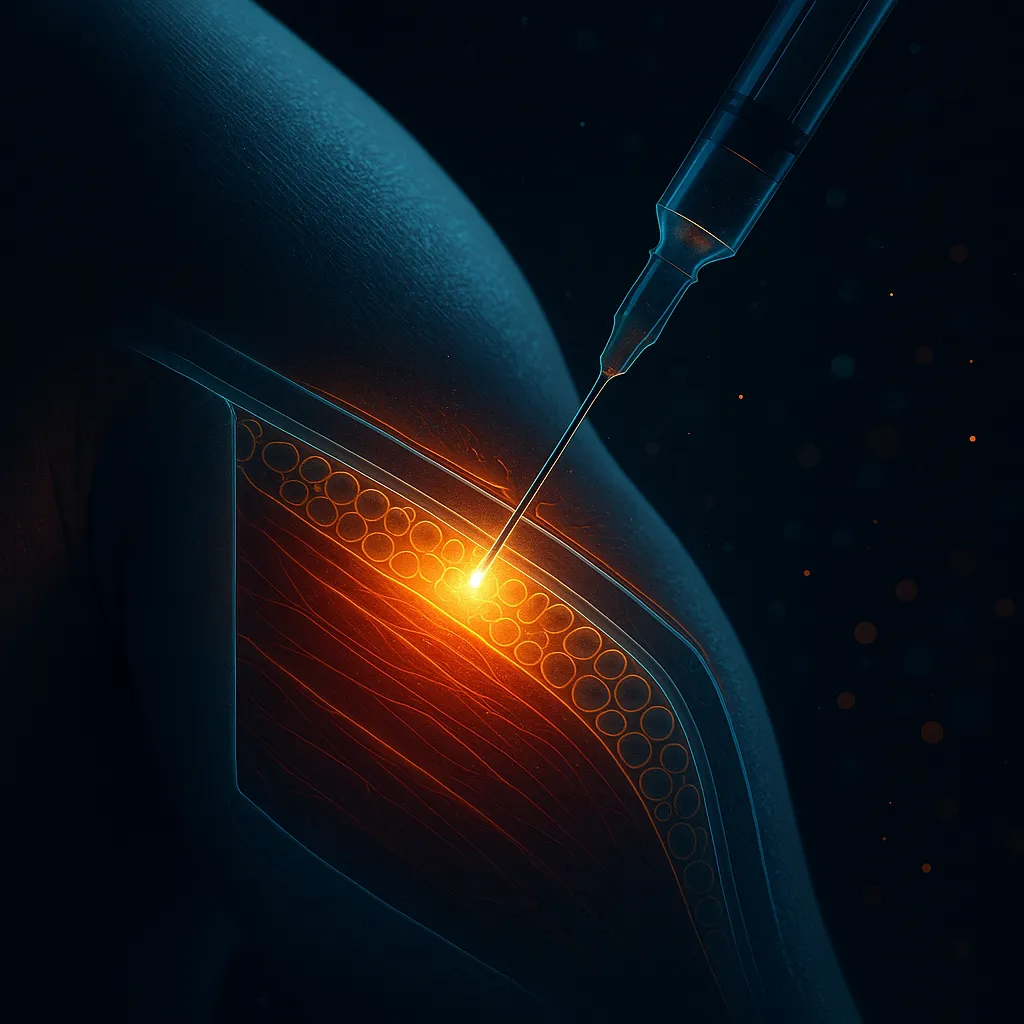
1. Injection Site Reactions
Frequency: Most users experience some degree Duration: 15-60 minutes post-injection
The most common side effect by far. Redness, mild swelling, and occasional itching at the injection site. This is a normal immune response to subcutaneous injection and not specific to the Wolverine Stack — it happens with most injectable peptides.
How to minimize:
- Rotate injection sites (4-6 abdominal spots, 2 inches from navel)
- Allow alcohol swab to dry completely before inserting needle
- Inject slowly and hold for 5 seconds before withdrawing
- Use 29-31 gauge needles (thinner = less trauma)
When to worry: If redness expands beyond 2 inches, feels hot to the touch, or persists more than 24 hours, this could indicate infection. Discontinue and consult a physician.
2. Mild Headache
Frequency: 10-20% of users, typically first week Duration: 1-4 hours, resolves spontaneously
BPC-157 modulates the nitric oxide (NO) system, which affects vascular tone. Some users experience transient headaches during the first few days as NO pathways adjust. This is similar to the headache mechanism seen with NO-boosting supplements.
How to minimize:
- Stay well hydrated
- Start at a lower dose (250mcg total vs 500mcg) for the first 3 days
- If persistent, reduce dose by half
Uncommon Side Effects (1-10% of Users)
3. Nausea
Frequency: 5-10%, almost always in the first 2-3 days Duration: 30-60 minutes post-injection
More common with higher starting doses (750mcg+) and when injecting on a full stomach. BPC-157 is a gastric peptide — derived from human gastric juice — and may modulate GI signaling during the initial adjustment period.
How to minimize:
- Inject on a mostly empty stomach (or wait 2+ hours after eating)
- Start at 250mcg and titrate up over 3-5 days
- If persistent, split the daily dose into two injections (morning and evening)
4. Fatigue or Drowsiness
Frequency: 5-10%, typically weeks 1-2 Duration: A few hours post-injection
Some users report mild drowsiness, particularly in the first week. The mechanism isn't well understood but may relate to BPC-157's interaction with dopaminergic and serotonergic systems, or the body's healing response requiring more rest.
How to minimize:
- Inject in the evening if drowsiness is problematic
- Ensure adequate sleep during the cycle (your body is repairing tissue)
5. Lightheadedness
Frequency: 3-5%, typically first few days Duration: Brief (minutes)
Possibly related to BPC-157's NO modulation affecting blood pressure transiently. More common in users who are already on blood pressure medication.
How to minimize:
- Inject while seated
- Wait 5-10 minutes before standing after injection
- Stay hydrated

Serious Concerns (Rare but Important)
6. Theoretical Cancer Risk (TB-500/Thymosin Beta-4)
Evidence level: Theoretical concern based on mechanism, not observed in practice
This is the most discussed safety concern with the Wolverine Stack, and it deserves a thorough, honest analysis.
The concern stems from TB-500's parent protein, thymosin beta-4, being found overexpressed in certain tumor types. In a mouse melanoma study, cells engineered to overexpress thymosin beta-4 produced significantly larger tumors with increased blood vessel formation compared to controls (Noh et al., 2003).
Thymosin beta-4 promotes angiogenesis — new blood vessel formation (Philp et al., 2004). This is beneficial for wound healing but could theoretically feed existing tumors by providing them with blood supply.
What the evidence actually shows:
- TB-500 has been found overexpressed in some tumors — but correlation does not equal causation
- No study has demonstrated that exogenous TB-500 causes cancer
- Some studies show thymosin beta-4 levels are actually lower in certain cancers (multiple myeloma)
- The overexpression studies used sustained, high-level genetic overexpression — very different from periodic subcutaneous injection
The balanced position: There is no evidence that Wolverine Stack use causes cancer. However, the angiogenic mechanism creates a theoretical risk for anyone with undiagnosed or existing malignancy. If you have active cancer, a recent cancer history, or strong family history, consult an oncologist before using TB-500 or the Wolverine Stack.
7. Allergic Reaction
Frequency: Very rare (<1%) Severity: Potentially serious
True allergic reactions to BPC-157 or TB-500 are extremely rare but possible with any injectable. Signs include:
- Widespread rash or hives beyond the injection site
- Swelling of face, lips, or throat
- Difficulty breathing
- Rapid heartbeat
Action: Discontinue immediately and seek emergency medical attention. This is not dose-dependent — if you have a true peptide allergy, even small amounts can trigger a reaction.