
On March 27, 2026, the Associated Press broke a story that ran across PBS, ABC, US News, and the Washington Times: dietary supplement makers are pushing the FDA to expand what qualifies as a supplement ingredient — and peptides are at the top of that list. The FDA held a public meeting to discuss whether its longstanding criteria for dietary supplements should be broadened to include substances that don't come from food, vitamins, herbs, or other traditional ingredients.
The political fight is interesting. But the science is more interesting.
Forget the lobbying for a moment. The real question is mechanical: do peptides actually work like supplements? Or is calling BPC-157 a "supplement" like calling a scalpel a butter knife because they're both made of metal?
The answer requires understanding how each category actually works at the molecular level — and that's where the conversation gets honest.
Top Retatrutide Vendors
Ranked by price, COA availability, and reputation
How Supplements Actually Work (Mechanistically)
Most supplements fall into a straightforward category: they provide raw materials or substrates that your body already uses. They don't instruct cells. They don't bind receptors with high specificity. They fill tanks.
Here are five familiar examples, broken down by their actual mechanism.
Creatine: The ATP Buffer
Creatine is the most well-studied supplement in existence, and its mechanism is beautifully simple. Your muscles store creatine as phosphocreatine — a molecule holding a high-energy phosphate group. When you need rapid energy (a heavy lift, a sprint), creatine kinase strips that phosphate and hands it to ADP, regenerating ATP in milliseconds.
That's it. No receptor binding. No signaling cascade. No gene expression changes. Creatine is a fuel reservoir. You're adding more phosphate donors to the tank so your muscles can regenerate ATP faster during high-intensity work.
This is why creatine works so reliably — it's a substrate, not a signal. Your body doesn't need to "interpret" creatine. It just uses it as raw material for the phosphocreatine shuttle [1].
Glutamine: The Nitrogen Shuttle
Glutamine is the most abundant amino acid in the body and the primary nitrogen shuttle between organs. It fuels enterocytes (gut lining cells), serves as substrate for immune cells during stress, and donates nitrogen for nucleotide synthesis.
Like creatine, glutamine is a building block. Supplementing it during illness or heavy training replaces what's been depleted. It doesn't activate a receptor cascade — it's metabolized as fuel by the cells that need it. Conditionally essential, not conditionally signaling.
Fish Oil (Omega-3s): Membrane Incorporation
Omega-3 fatty acids (EPA and DHA) get incorporated into cell membranes, altering membrane fluidity and serving as precursors for resolvins and protectins — molecules that help resolve inflammation. EPA and DHA also compete with arachidonic acid for enzymatic conversion, shifting the balance from pro-inflammatory to anti-inflammatory eicosanoids.
This is substrate-level activity. Fish oil doesn't bind a specific receptor and trigger a defined cascade. It changes the raw material composition of cell membranes, and downstream effects follow from that change. Important, but fundamentally different from receptor-targeted signaling.
Collagen Peptides: Where the Line Starts to Blur
Collagen is the supplement that makes this conversation complicated — and honest.
When you take hydrolyzed collagen, digestive enzymes break it into di- and tripeptides (primarily Gly-Pro-Hyp and Pro-Hyp). Unlike most dietary peptides, these fragments survive digestion and enter the bloodstream intact. Studies using radioactive labeling have confirmed these peptides reach the dermis, joints, and other connective tissues [2].
Here's where it gets interesting: collagen-derived dipeptides don't just serve as building blocks for collagen synthesis. Research shows Pro-Hyp can stimulate fibroblast proliferation and increase hyaluronic acid synthesis through activation of the TGF-beta1/Smad signaling pathway. That's not substrate behavior — that's signaling.
Collagen peptides sit in a gray zone. They're partially substrate (providing amino acids for collagen synthesis) and partially signal (triggering fibroblast activity through specific pathways). This gray zone is exactly where the supplement-vs-drug debate gets real.
Vitamin D: The "Supplement" That's Actually a Hormone
Vitamin D is sold on every supplement shelf in America, but mechanistically, it's a secosteroid hormone. After conversion to its active form (1,25-dihydroxyvitamin D), it binds the vitamin D receptor (VDR) — a nuclear receptor that directly modulates gene transcription. VDR activation influences over 200 genes involved in calcium metabolism, immune function, cell differentiation, and inflammation.
That's receptor binding. That's gene expression modulation. That's a signaling mechanism indistinguishable from what peptides do.
Vitamin D got grandfathered into the "supplement" category for historical reasons, not mechanistic ones. If vitamin D were discovered today and pitched to the FDA as a new ingredient, its receptor-targeted gene modulation would likely put it in the drug column. It's the exception that proves the rule — and the precedent that supplement makers point to when arguing peptides deserve the same treatment.
How Peptides Actually Work (Mechanistically)
Peptides don't fill tanks. They send instructions. The fundamental difference is specificity: a peptide binds a defined receptor, triggers a defined signaling cascade, and produces a defined biological response. It's the difference between delivering bricks to a construction site and sending an architect with blueprints.
BPC-157: Multi-Pathway Signaling
BPC-157 (Body Protection Compound-157) is a 15-amino-acid peptide derived from human gastric juice. Its mechanism has nothing to do with providing raw materials.
BPC-157 activates the Src-Caveolin-1-eNOS pathway, increasing nitric oxide production and promoting endothelial cell migration. It upregulates VEGF receptor-2 (VEGFR2), stimulating new blood vessel formation. It modulates the FAK-paxillin pathway involved in cell adhesion and tissue remodeling [3].
This is not substrate behavior. BPC-157 doesn't get metabolized for energy or incorporated into tissue as a building block. It binds to specific molecular targets and triggers cascading downstream effects — angiogenesis, cytoprotection, accelerated tendon and ligament repair. One molecule activating multiple signaling pathways is the textbook definition of a drug mechanism.
You don't "run out" of BPC-157 the way you run out of creatine. It's not a consumable. It's a message. For a deeper look at these healing mechanisms, see our BPC-157 benefits breakdown.
Semaglutide: High-Specificity Receptor Agonism
Semaglutide binds the GLP-1 receptor with 94% structural homology to the native GLP-1 hormone — but it's engineered to resist DPP-4 degradation, extending its half-life from minutes to approximately 7 days [4].
The downstream cascade is precise: GLP-1 receptor activation in the hypothalamus suppresses appetite by stimulating anorexigenic POMC/CART neurons and inhibiting orexigenic NPY/AgRP neurons. Peripherally, it slows gastric emptying and enhances glucose-dependent insulin secretion. Clinical trials (STEP program) demonstrated 15-17% average body weight loss.
There is no supplement on Earth that works this way. Semaglutide doesn't provide a nutrient your body is missing. It hijacks a specific receptor system with engineered precision to produce a specific outcome. That's a drug. Newer GLP-1 receptor agonists like retatrutide take this even further, hitting GLP-1, GIP, and glucagon receptors simultaneously — a triple mechanism no supplement could replicate. See how retatrutide compares to tirzepatide.
GHK-Cu: Gene Expression Modulation
GHK-Cu is a naturally occurring tripeptide (glycyl-L-histidyl-L-lysine) that binds copper and declines significantly with age. When researchers ran GHK through the Broad Institute's Connectivity Map, they found it modulates the expression of 4,048 human genes — roughly 6% of the genome [5].
These aren't random fluctuations. GHK-Cu upregulates genes involved in collagen synthesis, DNA repair, antioxidant defense, and anti-inflammatory signaling while downregulating genes associated with tissue destruction and inflammation. It promotes stem cell differentiation and increases secretion of trophic factors by mesenchymal stem cells.
A tripeptide that modulates 6% of the human genome is not a supplement. Creatine modulates zero genes. Fish oil alters membrane composition, which has downstream gene effects, but nothing approaching the scope or specificity of GHK-Cu's direct transcriptional influence.
TB-500: Actin Regulation and Cell Migration
TB-500 (a synthetic fragment of Thymosin Beta-4) is a 43-amino-acid peptide that sequesters G-actin monomers, regulating the assembly and disassembly of the actin cytoskeleton — the internal scaffolding that gives cells their shape and allows them to move [6].
By controlling actin dynamics, TB-500 enables cells to form lamellipodia and filopodia (the "feet" cells use to crawl). This accelerates cell migration toward injury sites, promotes angiogenesis through endothelial cell movement, and facilitates tissue remodeling. The seven-amino-acid actin-binding motif (LKKTETQ) is the functional core driving this activity.
No supplement does this. Glutamine doesn't reorganize your cytoskeleton. Fish oil doesn't direct cell migration. TB-500 tells cells where to go and gives them the structural tools to get there. Researchers often stack BPC-157 and TB-500 for complementary healing pathways — one drives angiogenesis while the other directs cell migration.
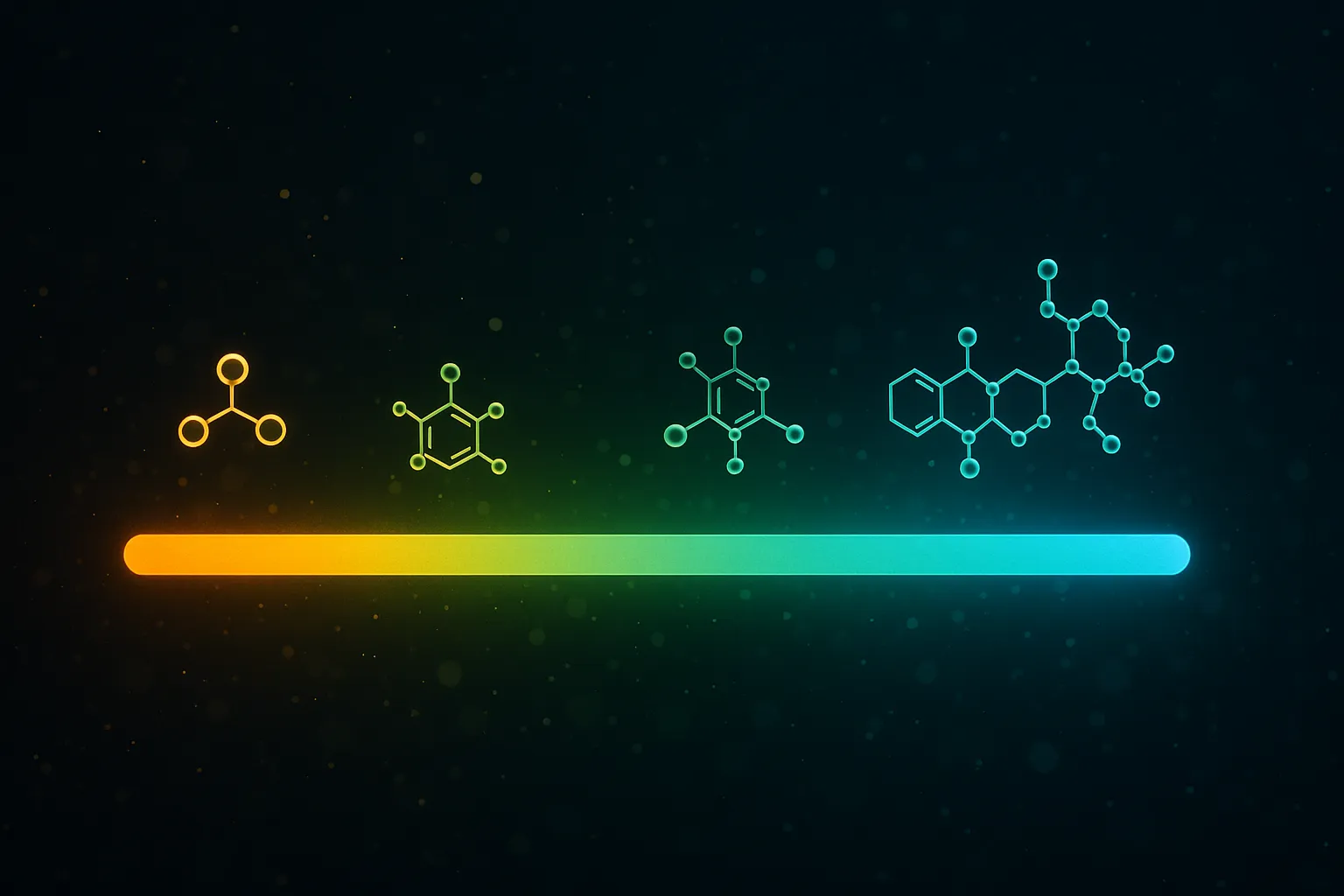
The Spectrum: Where the Line Actually Is
This isn't a binary. There's a spectrum from "definitely a supplement" to "definitely a drug," and understanding where each compound falls reveals why the FDA debate is so heated.
| Compound | Mechanism Type | Target Specificity | Receptor Binding | Gene Modulation | Classification |
|---|---|---|---|---|---|
| Creatine | Substrate (phosphate buffer) | None — metabolized as fuel | No | No | Supplement |
| Glutamine | Substrate (nitrogen donor) | None — metabolized as fuel | No | No | Supplement |
| Fish oil | Substrate (membrane incorporation) | Low — changes membrane composition | Indirect | Indirect, broad | Supplement |
| Collagen peptides | Substrate + partial signaling | Low-moderate — some fibroblast activation | Partial | Limited, pathway-specific | Supplement (gray zone) |
| Vitamin D | Receptor-targeted hormone | High — VDR nuclear receptor | Yes (VDR) | Yes (200+ genes) | Supplement (historical) |
| BPC-157 | Multi-pathway signaling | High — VEGFR2, eNOS, FAK-paxillin | Yes (multiple) | Yes (growth factors) | Drug mechanism |
| GHK-Cu | Gene expression modulator | Very high — 4,048 genes | Yes (copper-dependent) | Yes (6% of genome) | Drug mechanism |
| TB-500 | Actin regulation / cell migration | High — G-actin sequestering | Yes (actin-binding motif) | Yes (migration, repair) | Drug mechanism |
| Semaglutide | High-specificity receptor agonist | Very high — GLP-1R exclusive | Yes (GLP-1 receptor) | Yes (metabolic cascades) | Drug (FDA-approved) |
The pattern is clear. As you move from left to right, three things increase simultaneously:
- Target specificity — from "metabolized wherever needed" to "binds one receptor type"
- Signaling complexity — from "no cascade" to "multi-pathway downstream effects"
- Predictability of outcome — from "generally supports function" to "produces specific, measurable biological response"
Supplements occupy the left side. Peptides occupy the right. Collagen and vitamin D sit uncomfortably in the middle — and that middle ground is exactly what supplement makers are trying to exploit.
What the FDA Fight Is Actually About
The March 2026 AP story captures a collision between three powerful interests, and peptides are caught in the crossfire.
Supplement Makers Want Expanded Ingredient Lists
The FDA held a public meeting on March 28, 2026 to discuss whether its longstanding criteria for dietary supplements should be broadened. Currently, the Dietary Supplement Health and Education Act of 1994 (DSHEA) limits supplement ingredients to vitamins, minerals, herbs, amino acids, and substances "used to supplement the diet." Supplement companies argue this definition is outdated and should include peptides, probiotics, and other compounds that don't fit neatly into those categories.
Some companies have already been adding peptides to capsules, gummies, and powders. Technically, those products violate FDA rules. The public meeting was about whether to change the rules to match the market reality.
The Compounding Pharmacy Track
Separately, HHS Secretary Robert F. Kennedy Jr. announced on February 27, 2026 that approximately 14 of 19 peptides previously restricted from compounding will return to Category 1 status — making them available through licensed compounding pharmacies with a physician's prescription. This is a medical access pathway, not a supplement pathway [7].
The distinction matters. Category 1 compounding means:
- Physician prescription required
- Pharmaceutical-grade preparation from licensed pharmacies
- Individual patient formulations under medical oversight
- Quality testing and inspected facilities
This is fundamentally different from selling BPC-157 gummies at GNC.
The Real Tension
Three groups want peptide access, each through different channels:
Supplement companies want to sell peptides over the counter as dietary supplements — no prescription, no physician oversight, minimal regulatory burden. The March 2026 FDA meeting was about this pathway.
Compounding pharmacies want to prepare peptides under physician prescription — regulated but accessible. The Kennedy announcement addresses this pathway.
Pharmaceutical companies want peptides to go through full FDA approval — the most expensive and restrictive pathway, but the one that generates the most revenue and market exclusivity. Semaglutide went this route.
For consumers, the question is straightforward: which pathway gives you the best combination of access, quality, and safety? The mechanisms outlined above suggest that most bioactive peptides produce drug-like effects that warrant at least some level of medical oversight — even if full FDA approval is overkill for compounds like BPC-157 with decades of animal research and growing human safety data.
For a detailed breakdown of which peptides are returning to legal compounding status, see our FDA peptide reclassification guide.
Why This Matters for You
The classification debate isn't academic. How the FDA categorizes these compounds directly affects how you can access them, what quality guarantees you get, and what risks you're taking.
If Peptides Become Supplements
Lower barrier to access — you could buy BPC-157 capsules at a supplement store without a prescription. But supplement-grade manufacturing has dramatically less oversight than pharmaceutical-grade. The FDA does not review supplements for safety or efficacy before they hit shelves. Independent testing has repeatedly found supplements with incorrect doses, unlisted ingredients, or contamination.
For substrate supplements like creatine, this risk is manageable. Creatine is creatine — if the dose is slightly off, the consequences are minimal. For a compound like BPC-157 that activates VEGF pathways and modulates nitric oxide signaling, dosing precision matters significantly more.
If They Stay Classified as Drugs
Physician oversight provides a safety layer, but it also creates access barriers. Full FDA drug approval costs hundreds of millions of dollars and takes 10-15 years. No one is running Phase III trials on BPC-157 because the molecule can't be patented — there's no financial incentive to fund that approval process.
This is the access paradox: the regulatory pathway designed to ensure safety also ensures that many promising compounds never become legally available through conventional medical channels. For a full list of what's happened, see every peptide vendor that shut down in 2025-2026.
The Compounding Pharmacy Middle Ground
The Category 1 reclassification represents a pragmatic middle path. Licensed compounding pharmacies can prepare these peptides under physician supervision, providing pharmaceutical-grade quality without requiring full FDA approval. It's not perfect — compounded medications don't undergo the same level of clinical testing as FDA-approved drugs — but it's a significant improvement over both the grey market and the supplement aisle.
For most bioactive peptides, compounding with physician oversight is the approach that best matches their mechanistic profile: too targeted and potent for the supplement model, too well-studied for a blanket ban, not commercially viable for full FDA approval.